Fueling the Bioeconomy: Clean Energy Policies Driving Biotechnology Innovation
The transition to a clean energy future and diversified sources of energy requires a fundamental shift in how we produce and consume energy across all sectors of the U.S. economy. The transportation sector, a sector that heavily relies on fossil-based energy, stands out not only because it is the sector that releases the most carbon into the atmosphere, but also for its progress in adopting next-generation technologies when it comes to new technologies and fuel alternatives.
Over the past several years, the federal government has made concerted efforts to support clean energy innovation in transportation, both for on-road and off-road. Particularly, in hard-to-electrify transportation sub-sectors, there has been added focus such as through the Sustainable Aviation Fuel (SAF) Grand Challenge. These efforts have enabled a wave of biotechnology-driven solutions to move from research labs to commercial markets, such as LanzaJets alcohol-to-jet technology in producing SAF. From renewable fuels to bio-based feedstocks, biotechnologies are enabling the replacement of fossil-derived energy sources and contributing to a more sustainable, secure, and diversified energy system.
SAF in particular has gained traction, enabled in part by public investment and interagency coordination, like the SAF Grand Challenge Roadmap. This increased federal attention demonstrated how strategic federal action, paired with demand signals from government, targeted incentives, and industry buy-in, can create the conditions needed to accelerate biotechnology adoption.
To better understand the factors driving this progress, FAS conducted a landscape analysis at the federal and regional level of biotechnology innovation within the clean energy sector, complemented by interviews with key stakeholders. Several policy mechanisms, public-private partnerships, and investment strategies were identified that were enablers of advanced SAF adoption and production and similar technologies. By identifying the enabling conditions that supported biotechnology’s uptake and commercialization, we aim to inform future efforts on how to accelerate other sectors that utilize biotechnologies and overall, strengthen the U.S. bioeconomy.
Key Findings & Recommendations
An analysis of the federal clean energy landscape reveals several critical insights that are vital for advancing the development and deployment of biotechnologies. Federal and regional strategies are central to driving innovation and facilitating the transition of biotechnologies from research to commercialization. The following key findings and actionable recommendations address the challenges and opportunities in accelerating this transition.
Federal Level Key Findings & Recommendations
The federal government plays a pivotal role in guiding market signals and investment toward national priorities. In the clean energy sector, decarbonizing aviation has emerged as a strategic objective, with SAF serving as a critical lever. Federal initiatives such as the SAF Grand Challenge, the SAF Roadmap, and the SAF Metrics Dashboard have helped to elevate SAF within national climate priorities and enabled greater interagency coordination. These mechanisms not only track progress but also communicate federal commitment. Still, despite these efforts, current SAF production remains far below target levels, with capacity largely concentrated in HEFA, a pathway with constrained feedstock availability and limited scalability.
This production gap reflects deeper structural challenges, many of which parallel broader issues across the clean-energy biotech interface. One of the main challenges is the fragmented, short-duration policy incentives currently in use. Tax credits like 40B and 45Z, while important, lack the longevity and clarity required to unlock large-scale, long-term private investment. The absence of binding fuel mandates further undermines market certainty. These policy gaps limit the ability of the clean energy sector to serve as a sustained demand signal for emerging biotechnologies and slow the transition from pilot to commercial scale.
Importantly, these challenges point to a broader opportunity: SAF as a test case for how the clean energy sector can serve as a driver of biotechnology uptake. Promising biotechnologies, such as alcohol-to-jet and power-to-liquid, are currently stalled by high capital costs, uncertain regulatory pathways, and a lack of coordinated federal support. Addressing these bottlenecks through aligned incentives, technology-neutral mandates, and harmonized accounting frameworks could not only accelerate SAF deployment but also establish a broader policy blueprint for scaling biotechnology across other clean energy applications.
To alleviate some of the challenges identified, the federal government should:
Extend & Clarify Incentives
While tax incentives such as the 45Z Clean Fuel Production Credit offer a promising framework to accelerate low-carbon fuel deployment, current design and implementation challenges limit their impact, particularly for emerging bio-based and synthetic fuels. To fully unlock the climate and market potential of these incentives, Congress and relevant agencies should take the following steps:
- Congress should amend the 45Z tax credit structure to differentiate between fuel types, such as SAF, e-fuels, biofuels, and renewable diesel, based on life cycle CI and production pathways. This would better reflect technology-specific costs and accelerate deployment across multiple clean fuel markets, adding specificity as to how to utilize and earn the credits based on the type of fuel.
- Congress should extend the duration of the 45Z credit and other clean-fuel related incentives to provide long-term policy certainty. Multi-year extensions with a defined minimum value floor would reduce investment risk and enable financing of capital-intensive projects.
- Congress and the Department of Treasury should clarify eligibility to ensure inclusion of co-processing methods and hybrid production systems, which are currently in regulatory gray areas. This would ensure broader participation by innovative fuel producers.
- Federal agencies, including the Department of Energy (DOE), Department of Transportation (DOT), and the Department of Defense, should be directed to enter into long-term (more than 10 years) procurement agreements for low-carbon fuels, including electrofuels and SAF. These offtake mechanisms would complement tax incentives and send strong market signals to producers and investors.
Scale Biotech Commercialization Support
The clean energy transition depends in part on the successful commercialization of enabling biotechnologies, ranging from advanced biofuels to bio-based carbon capture, SAF and biomanufacturing platforms that reduce industrial emissions. Recent or proposed funding cuts to clean energy programs risk stalling this progress and undermining U.S. competitiveness in the bioeconomy.
To accelerate biotechnology deployment and bridge the gap between lab-scale innovation and commercial-scale production, Congress should take the following actions:
- Authorize and appropriate expanded funding to the DOE, particularly through Bioenergy Technologies Office (BETO) and to the Department of Agriculture (USDA) to support pilot, demonstration, and first-of-a-kind commercial scale projects that enable biotechnology applications across clean energy sectors.
- Direct and fund the DOE Loan Programs Office to establish a dedicated loan guarantee program focused on biotechnology commercialization, targeting platforms that can be integrated into the energy system, such as bio-based fuels, bioproducts, carbon utilization technologies, and electrification-enabling materials.
- Encourage DOE and USDA to enter into long-term offtake agreements or structured purchasing mechanisms with qualified bioenergy and biomanufacturing companies. These agreements would help de-risk early commercial projects, crowd in private investment, and provide market certainty during the critical scale-up phase.
- Strengthen public-private coordination mechanisms, such as cross-sector working groups or interagency task forces, to align commercialization support with industry needs, improve program targeting, and reduce time-to-market for promising technologies.
Design and Promote Next-Gen Biofuel Policies
To accelerate the deployment of low-carbon fuels and enable innovation in next-generation bioenergy technologies, Congress and relevant agencies should take the following actions:
- Congress should direct the Environmental Protection Agency (EPA) to modernize the Renewable Fuel Standard by incorporating life cycle carbon intensity as a core metric, moving beyond volume-based mandates. Legislative authority could also support the development of a national Low Carbon Fuel Standard, modeled on successful state-level programs to drive demand for fuels with demonstrable climate benefits.
- EPA should update its emissions accounting framework to reflect the latest science on life cycle greenhouse gas (GHG) emissions, enabling more accurate assessment of advanced biofuels and synthetic fuels.
- DOE should expand R&D and demonstration funding for biofuel pathways that meet stringent carbon performance thresholds, with an emphasis on scalability and compatibility.
Regional Level Key Findings & Recommendations
Regional strengths continue to serve as foundational drivers of clean energy innovation, with localized assets shaping the pace and direction of technology development. Federal designations, such as the Economic Development Administration (EDA) Tech Hub program (Tech Hub), have proven catalytic. These initiatives enable regions to unlock state-level co-investment, attract private capital, and align workforce training programs with local industry needs. Early signs suggest that the Tech Hub framework is helping to seed innovation ecosystems where they are most needed, but long-term impact will depend on sustained funding support and continued regional coordination.
Workforce readiness and enabling infrastructure remain critical differentiators. Regions with deep and committed involvement from major research universities, national labs, or advanced manufacturing clusters are better positioned to scale innovation from prototype to deployment. Real-world testbeds provide environments for stress-testing technologies and accelerating regulatory and market readiness, reinforcing the importance of place-based strategies in federal innovation planning.
At the same time, private investment in clean energy and enabling biotechnologies remains crucial to developing and scaling innovative technologies. High capital costs, regulatory uncertainty, and limited early-stage demand signals continue to inhibit market entry, especially in geographies with less mature innovation ecosystems. Addressing these barriers through coordinated federal procurement, long-term incentives, and regional capacity-building will be essential to supporting growth in regions with strong assets to develop industry clusters that could yield clean energy benefits.
To accomplish this, the federal government and regional governments should:
Strengthen Regional Workforce Pipelines
A skilled and regionally distributed workforce is essential to realizing the full economic and technological potential of clean energy investments, particularly as they intersect with the bioeconomy. While federal funding is accelerating deployment through initiatives such as the IRA and DOE programs, workforce gaps, especially outside major innovation hubs, pose barriers to implementation. Addressing these gaps through targeted education, training, and talent retention efforts will be critical to ensuring that clean energy projects deliver durable, regionally inclusive economic growth. To this end:
- Federal agencies like the Department of Education and National Science Foundation should explore expanding support for STEM programs at community colleges and Minority Serving Institutions, with a focus on biosciences, engineering, and agricultural technologies relevant to the clean energy transition.
- Federally supported training and reskilling programs tailored to regional clean energy and biomanufacturing workforce needs could benefit new and existing cross-sector partnerships between state workforce agencies and regional employers.
- State and local governments should consider implementing talent retention strategies, including local hiring incentives, relocation support, and career placement services, to ensure that skilled workers remain in and contribute to regional clean energy ecosystems.
Strengthen Regional Infrastructure and Foster Cross-Sector Collaboration
Robust regional infrastructure and cross-sector collaboration are essential to accelerating the deployment of clean energy technologies that leverage advancements in biotechnology and manufacturing. Strategic investments in shared facilities, modernized logistics, and coordinated innovation ecosystems will strengthen supply chain resilience and improve technology transfer across sectors. Facilitating access to R&D infrastructure, particularly for small and mid-sized enterprises, will ensure that innovation is not limited to large firms or major metropolitan areas. To support these outcomes:
- Federal support for regional testbeds, prototyping sites, and grid modernization labs, coordinated by agencies such as DOE and EDA, would support the demonstration and scaling of biologically enabled clean energy technologies.
- State and local governments, in coordination with federal agencies including DOT and DOC, explore investment in logistics infrastructure to enhance supply chain reliability and support distributed manufacturing.
- States should consider creating or expanding the use of innovation voucher programs that allow small and mid-sized enterprises to access national lab facilities, pilot-scale infrastructure, and technical expertise, fostering cross-sector collaboration between clean energy, biotech, and advanced manufacturing firms.
Attract and De-Risk Private Capital
Attracting and de-risking private capital is critical for scaling clean energy and biotechnology innovations. By offering targeted financial mechanisms and leveraging federal visibility, governments can reduce the financial uncertainties that often deter private investment. Effective strategies, such as state-backed loan guarantees and co-investment models, can help bridge funding gaps while strategic partnerships with philanthropic and venture capital entities can unlock additional resources for emerging technologies. To this end:
- State governments, in collaboration with the federal agencies such as DOE and Treasury, should consider implementing state-backed loan guarantees and co-investment models to attract private capital into high-risk clean energy and biotech projects.
- Federal agencies like EDA, DOE, and the SBA should explore additional programs and partnerships to attract philanthropic and venture capital to emerging clean energy technologies, particularly in underserved regions.
- Federal agencies should increase efforts to facilitate connecting early-stage companies with potential investors, using federal initiatives to build investor confidence and reduce perceived risks in the clean energy sector.
Cross-Cutting Key Findings
The successful deployment of federal clean energy and biotechnology initiatives, such as the SAF Grand Challenge, relies heavily on the capacity of regional ecosystems and the private sector to absorb and implement national goals. Many regions, particularly those outside established innovation hubs, lack the infrastructure, resources, and technical expertise to effectively utilize federal funding. As a result, the impact of national policies is often limited, and the full potential of federal investments goes unrealized in certain areas.
Federal programs often take a one-size-fits-all approach, overlooking regional variability in feedstocks, industrial bases and cost structures. Programs like tax credits and life cycle analysis models can unintentionally disadvantage regions with different economic contexts, creating disparities in access to federal incentives. This lack of regional customization prevents certain areas from fully benefiting from national clean energy and biotech initiatives.
The diffusion of innovation in clean energy and biotechnology remains concentrated in a few key regions, leaving others underutilized. Despite robust federal R&D investments, commercialization and scaling of innovations are primarily concentrated in regions with established infrastructure, hindering the broader geographic spread of these technologies. In addition, workforce development efforts across federal and regional programs are fragmented, creating misalignments in talent pipelines and further limiting the ability of local industries to leverage available resources effectively. The absence of a unified system for tracking key metrics, such as SAF production and emissions reductions, makes it difficult to coordinate efforts or assess progress consistently across regions. To address this, the federal and regional governments should:
Create a Federal–Regional Clean Energy Deployment Compact
A Federal-Regional Clean Energy Deployment Compact is critical for aligning federal clean energy initiatives with the unique capabilities and needs of regional ecosystems. By establishing formal mechanisms, such as intergovernmental councils and regional liaisons, federal programs can be more effectively tailored to local conditions. These mechanisms will ensure two-way communication between federal agencies and regional stakeholders, fostering a collaborative approach that adapts to evolving technological, economic, and environmental conditions. In addition, treating regional tech hubs and initiatives as testbeds for new policy tools, such as performance-based incentives or carbon standards, will allow for innovative solutions to be tested locally before scaling them nationally, ensuring that policies are effective and contextually relevant across diverse regions. To this end:
- The White House Office of Science and Technology Policy (OSTP), in collaboration with the DOE and EPA, should establish formal intergovernmental councils or regional liaisons to facilitate ongoing dialogue between federal agencies and regional stakeholders. These councils would focus on aligning federal clean energy initiatives with regional needs, ensuring that local priorities, such as feedstock availability or infrastructure readiness, are considered in policy design.
- The DOE and EPA should treat tech hubs and regional clean energy initiatives as testbeds for policy innovation. These regions can pilot performance-based incentives, carbon standards, and other policy tools to assess their effectiveness before scaling them nationally. Successful models developed in these testbeds should be expanded to other regions, with lessons learned shared across state and local governments.
- Regional universities and innovation hubs should collaborate with federal and state agencies to develop pilot programs that test new policy tools and technologies. These institutions can serve as incubators for innovative clean energy solutions, providing valuable data on what policies work best in specific regional contexts.
Build a National Innovation-to-Deployment Pipeline
Creating a seamless innovation-to-deployment pipeline is essential for scaling clean energy technologies and ensuring that regional ecosystems can fully participate in national clean energy transitions. By linking DOE national labs, Tech Hubs, and regional consortia into a coordinated network, the U.S. can support the full life cycle of innovation, from early-stage R&D to commercialization and deployment, across diverse geographies. Additionally, co-developing curricula and training programs between federal agencies, regional tech hubs, and industry partners will ensure that talent pipelines are closely aligned with the evolving needs of the clean energy sector, providing the skilled workforce necessary to implement and scale innovations effectively. To accomplish this the:
- DOE should facilitate the creation of a national network that connects federal labs, regional tech hubs and innovation consortia. This network would provide a clear pathway for the transition of technologies from research to commercialization, ensuring that innovations can be deployed across different regions based on local needs and capacities.
- Regional Tech Hubs, in partnership with local universities and research institutions, should be integrated into the pipeline to provide localized innovation support and commercialization expertise. These hubs can act as nodes in the broader network, offering the infrastructure and expertise necessary for scaling up clean energy technologies.
Develop a Shared Metrics and Monitoring Platform
A centralized dashboard for tracking key metrics related to clean energy and biotechnology initiatives is crucial for guiding investment and policy decisions. By integrating federal and regional data can provide a comprehensive, real-time view of progress across the country. This shared platform would enable better coordination among federal, state, and local agencies, ensuring that resources are allocated efficiently and that policy decisions are informed by accurate, up-to-date data. Moreover, a unified system would allow for more effective tracking of regional performance, enabling tailored solutions and based on localized needs and challenges. To this end:
- The DOE, in partnership with the EPA, should lead the development of a centralized dashboard that integrates existing federal and regional data on SAF production, emissions reductions, workforce needs, and infrastructure gaps. This platform should be publicly accessible, allowing stakeholders at all levels to monitor progress and identify opportunities for improvement.
- State and local governments should contribute relevant data from regional initiatives, including workforce training programs, infrastructure development projects, and emissions reductions efforts. These contributions would help ensure that the platform reflects the full range of activities across different regions, providing a more accurate picture of national progress.
The Department of Labor and the DOC should integrate workforce development and industrial capacity data into the platform. This would include information on training programs, regional workforce readiness, and skills gaps, helping policymakers align talent development efforts with regional needs.
The DOT should ensure that transportation infrastructure data, particularly related to SAF production and distribution networks, is included in the platform. This would provide a comprehensive view of the supply chain and infrastructure readiness necessary to scale clean energy technologies across regions.
The clean energy sector, and specifically SAF, highlights both the promise and the persistent challenges of scaling biotechnologies, reflecting broader issues, such as fragmented regulation, limited commercialization support, and misaligned incentives that hinder the deployment of advanced biotechnologies. Overcoming these systemic barriers requires coordinated, long-term policies including performance-based incentives, and procurement mechanisms that reduce investment risk and free up capital. SAF should be seen not as a standalone initiative but as a model for integrating biotechnology into industrial and energy strategy, supported by a robust innovation pipeline, expanded infrastructure, and shared metrics to guide progress. With sustained federal leadership and strategic alignment, the bioeconomy can become a key pillar of a low-carbon, resilient energy future.
Translating Vision into Action: FAS Commentary on the NSCEB Final Report and the Future of U.S. Biotechnology
Advancing the U.S. leadership in emerging biotechnology is a strategic imperative, one that will shape regional development within the U.S., economic competitiveness abroad, and our national security for decades to come. In the past few years, the contribution of biotechnology to the U.S. economy (referred to as the bioeconomy) has grown significantly, contributing over $210 billion to GDP and creating more than 640,000 domestic jobs, cementing its role as a major and expanding economic force. The impact of biotechnologies and biomanufacturing can be seen across diverse sectors and geographies, with applications spanning agriculture, energy, industrial manufacturing, and health. As biotechnology continues to drive innovation, it is emerging as a core engine of the next industrial revolution.
To maximize the strategic potential of emerging biotechnology, Congress established the bipartisan National Security Commission on Emerging Biotechnology (NSCEB) through the FY22 National Defense Authorization Act. The commission was tasked with conducting a comprehensive review of how advancements in biotechnology and related technologies will shape the current and future missions of the Department of Defense (DOD), and developing actionable policy recommendations to support the adoption and advancement of biotechnology within DOD and across the federal government. This effort culminated in a report, “Charting the Future of Biotechnology”, delivered to Congress in April 2025. The final report outlines 49 recommendations aimed at accelerating biotechnology innovation and scaling the U.S. biomanufacturing base, reinforcing the bioeconomy as a strategic pillar of national security and economic competitiveness.
In addition to developing a series of recommendations to promote and grow the U.S. bioeconomy, the Commission has also been tasked with facilitating the implementation and adoption of these policy recommendations by Congress and relevant federal agencies. To date, several pieces of legislation have been introduced in both the 118th and 119th Congress that incorporate recommendations from NSCEB’s interim and final reports (Table 1). This Legislation Tracker will be updated as this legislation moves through the process and as new bills are introduced.
The NSCEB report represents a critical policy opportunity for the U.S. bioeconomy. It proposes an injection of $15 billion to support sustained growth in biotechnology innovation and biomanufacturing through strategic investment and improved coordination. This level of investment is significant and would signal congressional support for the bioeconomy that goes beyond that seen in CHIPS and Science Act and the Inflation Reduction Act of 2022. This much needed infusion of federal investment offers a timely opportunity to build on existing momentum and unlock the next phase of U.S. leadership in the bioeconomy.
The recommendations in the report should be seen as opportunities for engagement with the Commission and with Congress for further refinement of these policy ideas. As the Commission begins its work on implementation, they have called on stakeholders across the bioeconomy to help refine and strengthen its proposals. Responding to this need, the Federation of American Scientists (FAS) has identified priority areas requiring greater clarity and has issued an open call for supplemental recommendations and policy proposals through the Day One Open Call process.
Overall, FAS supports the Commission’s final report and we applaud the Commission’s efforts to elevate the national conversation around emerging biotechnologies. The report provides a necessary foundation for long-term federal strategy and investment in biotechnology and biomanufacturing. At the same time, there remain clear opportunities to strengthen the recommendations through greater specificity and deeper stakeholder engagement. Two overarching decisions by the Commission deserve some additional scrutiny. First, the report’s adversarial framing towards China, while grounded in strategic reality, risks overlooking opportunities for targeted collaboration that could yield global benefits, particularly in areas where scientific progress depends on multinational cooperation. Second, the final report gives limited attention to the agricultural sector, despite its clear relevance to national security and the DOD’s growing interest in agricultural biotechnology. The “Additional Considerations” section does include a constructive call to modernize the USDA’s BioPreferred Program and update federal classification systems, recommendations that echo those issued by FAS. A more comprehensive approach toward this sector is needed.
The following sections summarize the report’s key pillars and provide analysis, highlighting core recommendations and identifying opportunities where additional detail and stakeholder input, through the Day One Open Call, will be essential for translating the report’s vision into actionable, high-impact policy. Additionally, the Supplementary Recommendations Table for the NSCEB Final Report (Table 2) lists each of the recommendations from the pillars and cross references related proposals from prior FAS work, subject matter experts, and Day One Memos submitted by external stakeholders.
Table 2: Supplementary Recommendations Table for the NSCEB Final Report
Pillar 1. Prioritize Biotechnology at the National Level
Pillar 1 of the report emphasizes the need to prioritize biotechnology at the national level. The recommendations within this pillar are essential for the development of a cohesive national strategy, and we encourage Congress to consider incorporating terminology and drawing on previous policy related to the bioeconomy to ensure that previous progress related to emerging biotechnologies is not lost.
A central recommendation within this Pillar is the establishment of the National Biotechnology Coordination Office (NBCO), which would reduce fragmentation and elevate biotechnology as a national priority. To succeed, the NBCO must address challenges faced by past coordination bodies and be empowered by the administration to drive cross-agency strategy despite differing institutional perspectives. While the presidential appointment of the director could lend authority, it also risks politicization and strategic shifts that may destabilize the sector. Success will depend on clarity of roles, coordination across functions, and strong institutional support for implementation.
FAS provided several additional recommendations and insights on these topics (see Table 2) to make them more nuanced and actionable by Congress, including:
- Coordinating the U.S. Government Approach to the Bioeconomy by Sarah Carter
- A National Bioeconomy Manufacturing and Innovation Initiative by Alexander Titus
Pillar 2. Mobilize the Private Sector to get U.S. Products to Scale
Pillar 2 of the final report focuses on mobilizing the private sector to strengthen biotechnology products by addressing key challenges to the sector, including regulatory reform, financing obstacles, and infrastructure and data needs. While the report correctly identifies long standing regulatory bottlenecks for products of biotechnology under the Coordinated Framework, including unclear oversight and interagency conflicts, it also acknowledges statutory complexities that make reform difficult. Empowering the Office of Management and Budget’s Office of Information and Regulatory Affairs (OIRA) to mediate these disputes is a promising approach, but would require statutory reinforcement. Similarly, proposals to modernize regulatory capacity, such as agency fellowships and regulatory science programs, highlight a critical need for technical expertise within government, though questions remain about institutional placement and long-term sustainability.
On financing and infrastructure, the report points to real gaps in early-stage capital and scale-up capacity, particularly for bridging the “Valley of Death” for biotechnology manufacturing. Concepts like advance market commitments and a new investment fund have potential, but their impact will depend heavily on design, risk management, and alignment with existing capital pipelines. The infrastructure recommendations are strong, but coordination challenges, particularly among national labs, regional hubs, and entities like BioMADE, must be addressed to avoid duplication or underutilization and approaches to securing bioeconomy infrastructure and data are underdeveloped. It will be critical to better define what constitutes critical biotechnology infrastructure and how it should be protected.
FAS provided significant expertise on these topics (see Table 2), such as:
- Coordinating the U.S. Government Approach to the Bioeconomy by Sarah Carter
- Regulations for the Bioeconomy by Sarah Carter
- A National Frontier Tech Public-Private Partnership to Spur Economic Growth by Katie Rae, Orin Hoffman & Michael Kearney
- De-Risking the U.S. Bioeconomy by Establishing Financial Mechanisms to Drive Growth and Innovation by Nazish Jeffery & Zak Weston
- Advancing the U.S. Bioindustrial Production Sector by Michael Fisher
- Closing Critical Gaps from Lab to Market by Phil Weilerstein, Shaheen Mamawala, and Heath Naquin
- Strengthening the U.S. Biomanufacturing Sector Through Standardization by Chris Stowers
- Summary Report – December 7, 2022, Bioeconomy Policy Workshop: Financial and Economic Tools
- Project BOoST: A Biomanufacturing Test Facility Network for Bioprocess Optimization, Scaling, and Training by Ed Charles & Chris Fracchia
One of the NSCEB’s recommendations in particular would benefit from additional input from external subject matter experts to make it more concrete and actionable for Congress:
- Recommendation 2.2d: Congress should improve the effectiveness and reach of the Small Business Innovation Research (SBIR) and Small Business Technology Transfer (STTR) programs to support early-stage innovation. Specifically, stakeholder input on:
- Which areas of biotechnology or sectors within the bioeconomy would most benefit from SBIR and STTR investment?
- How can these programs better support not only early- but also late-stage innovation?
If you have specific policy suggestions related to this topic, we encourage you to submit your ideas through the Day One Open Call page at FAS.
Pillar 3. Maximize the Benefits of Biotechnology for Defense
Pillar 3 of the final report focuses on maximizing the benefits of biotechnology for national defense, with an emphasis on the intelligence community and the Department of Defense (DOD). This pillar includes recommendations related to BioMADE oversight as well as internal workforce education on biotechnology. While oversight of BioMADE is important, it is unclear why additional oversight from Congress is needed for this manufacturing institute above and beyond that provided by its federal sponsor. Additionally, it will be essential for DOD to establish a mechanism for regularly updating workforce education in biotechnology, given the sector’s rapid and continuous evolution.
More broadly, this Pillar appropriately reframes biotechnology as a strategic capability, beyond its traditional role in R&D. This shift in perspective is timely, but realizing the potential of these technologies will require significant institutional change. Ethical use frameworks, particularly around dual-use risks (warfighter enhancement, surveillance, and environmental impact) must be developed through a transparent process that extends beyond DOD to include external stakeholders and independent organizations. In addition, proposed investment and export controls aimed at limiting adversarial advantage must be carefully scoped and implemented. The Department of Commerce (DOC) has published multiple Requests for Information (2018, 2020) to understand and delineate “high-risk” biotech products. Yet, DOC has not added new biotech products to the export list, which highlights the complexity of this task, and underscores the need for precision to avoid stifling beneficial collaboration or disrupting global supply chains.
FAS provided several additional recommendations and insights on these topics (see Table 2) to make them more nuanced and actionable by Congress, including:
- CLimate Improvements through Modern Biotechnology (CLIMB) — A National Center for Bioengineering Solutions to Climate Change and Environmental Challenges by Jennifer Panlilio & Hanny Rivera
- A Foundational Technology Development and Deployment Office to Create Jobs by Katie Rae, Michael Kearney, and Orin Hoffman
Pillar 4. Out-Innovate our Strategic Competitors
Pillar 4 of the NSCEB’s report offers recommendations for strengthening the biotechnology sector to out-innovate global competitors. It focuses on building robust data ecosystems, enhancing biosecurity, and expanding bio R&D infrastructure within the U.S. A central theme is the creation of a modern biological data ecosystem, which would provide the foundational infrastructure necessary to accelerate innovation. While a biological data ecosystem and associated standards are timely, several technical and governance challenges must be addressed, like harmonizing legacy systems, defining AI-readiness, and coordinating cloud lab integration. These complexities present an opportunity for stakeholder engagement and thoughtful design.
Within the Pillar, proposals that call for expanding National Lab capabilities and funding interdisciplinary biotechnology research are directionally strong, but success will depend on interagency coordination and alignment with industry needs. Finally, the report calls for stronger governance of biosafety and biosecurity, though its assertion that past efforts have “failed” could benefit from more nuanced analysis.
While FAS provided expertise on these topics (see Table 2), such as:
- A National Bioeconomy Manufacturing and Innovation Initiative by Alexander Titus
- Kickstarting Collaborative, AI-Ready Datasets in the Life Sciences with Government-funded Projects by Erika DeBenedictis, Ben Andrew, and Pete Kelly
- Creating a National Exposome Project by Gurdane Bhutani, Gary Miller, and Sandeep Patel
- Accelerating Biomanufacturing and Producing Cost-Effective Amino Acids through a Grand Challenge by Allison Berke
- Accelerating Bioindustry Through Research, Innovation, and Translation by Jon Roberts
A few of the report’s recommendations would benefit from additional stakeholder input to enhance clarity and ensure they are actionable for Congress:
- Recommendation 4.1c: Congress should authorize and fund the Department of Interior to create a Sequencing Public Lands Initiative to collect new data from U.S. public lands that researchers can use to drive innovation and Recommendation 4.1d: Congress should authorize the National Science Foundation to establish a network of “cloud labs,” giving researchers state-of-the-art tools to make data generation easier. Specifically, stakeholder input on:
- What type of data should be generated and what types of data generation or collection should be prioritized?
- How can we best draw on or expand existing cloud lab capabilities?
- Recommendation 4.3b: Congress should initiate a grand research challenge focused on making biotechnology predictably engineerable. Specifically, stakeholder input on:
- What specific grand challenge should Congress pursue and how should it be implemented?
- How should the U.S. government engage the scientific community (and others) in establishing and pursuing grand challenges for biotechnology?
- Recommendation 4.4a: Congress must direct the executive branch to advance safe, secure, and responsible biotechnology research and innovation. Specifically, stakeholder input on:
- The report calls for establishment of a body within the U.S. government for this purpose. What should this look like and how would it operate?
If you have specific policy suggestions related to these topics, we encourage you to submit your ideas through the Day One Open Call at FAS.
Pillar 5. Build the Biotechnology Workforce of the Future
Pillar 5 of the final report looks to the future by offering recommendations to secure and build the biotechnology workforce needed in the future. It addresses both the modernization of the federal biotech workforce and the development of the broader U.S. biotech workforce. Modernizing the federal workforce requires more than training programs. It requires coordination across HR systems, consistent standards, and better integration of biotechnology experts into national security and diplomacy. Proposals to expand Congressional science capacity are long overdue and necessary to equip lawmakers to address rapidly evolving biotechnology issues. On the national level, scaling the biomanufacturing workforce will depend on aligning credentials with industry needs and securing input from labor, academia, and employers. Expanding biotechnology education is promising, but successful implementation will require investment in teacher training and curriculum development.
While FAS contributed several recommendations to support this critical capacity-building effort (see Table 2), such as:
- Gathering Industry Perspectives on how the U.S. Government can Support the Bioeconomy by Sarah Carter
- Meeting Biology’s “Sputnik Moment”: A Plan to Position the United States as a World Leader in the Bioeconomy by Natalie Kuldell
One of the NSCEB’s recommendation would benefit from additional stakeholder input to enhance its clarity and make it more actionable for Congress:
- Recommendation 5.2a: Congress must maximize the impact of domestic biomanufacturing workforce training programs. Specifically, stakeholder input on:
- How should the government approach creating competency models for biomanufacturing training and microcredentialing?
- Which specific areas are best suited for microcredentialing efforts?
If you have specific policy suggestions related to these topics, we encourage you to submit your ideas through the Day One Open Call at FAS.
Pillar 6. Mobilize the Collective Strengths of our Allies and Partners
Pillar 6 of the final report focuses on strengthening alliances and partnerships on the global stage to enhance the U.S. biotechnology sector. It highlights the role of the State Department (DOS) in facilitating this effort through development of foreign policy tools, strengthening global data and market infrastructure, and leading in the establishment of international standards within the sector. Elevating biotechnology within U.S. foreign policy is both timely and necessary, particularly as biotech becomes increasingly strategic in areas like health security, climate resilience, and defense. Leveraging existing tools like the International Technology Security and Innovation (ITSI) Fund could provide a solid foundation, but effective execution will require clearer interagency coordination, transparency in funding allocation, and defined metrics for impact, especially across overlapping technology domains.
Proposals to create shared data infrastructure, joint purchasing mechanisms, and international fellowships point to smart long-term strategies for building trust and interoperability with allies. Yet, success hinges on careful coordination, especially around sensitive areas like dual-use biotechnology export controls. If U.S. standards are significantly more restrictive than those of allies, it could create friction and undermine broader goals of international collaboration and leadership.
FAS provided several additional recommendations and insights on these topics (see Table 2) to make them more nuanced and actionable by Congress, including:
- A Matter of Trust: Helping the Bioeconomy Reach Its Full Potential with Translational Governance by Christopher J. Gillespie
- Strategic Investments the U.S. Should Make in the Bioeconomy Right Now by Nazish Jeffery
- Strengthening U.S. Engagement in International Standards Bodies by Natalie Thompson and Mark Montgomery
- What’s Next for the U.S. Bioeconomy? Defining It. by Nazish Jeffery
One particularly important recommendation emphasizes the need to engage the public and build trust in the sector by collecting data on public acceptance. This data can help inform national governance and ensure it is more responsive and translatable to public concerns.
Additional Considerations
The additional considerations section of the NSCEB report brings several key recommendations that do not fit with the rest of the report, though are still very important. Many focus on aligning federal leadership and economic infrastructure with the needs of a growing and strategically vital biotechnology sector. Elevating biotechnology leadership within DOD is a logical step to align R&D with budget authority and operational needs. Similarly, expanding the scope of the Bioenergy Technologies Office (BETO) beyond biofuels and codifying the Office of Critical and Emerging Technology (OCET) role reflects an overdue shift toward recognizing biotech’s relevance to national security and broader innovation policy, though these changes will require institutional buy-in and cultural adjustment. On the economic side, proposals to create a public-private innovation consortium are timely, especially for supporting smaller firms and navigating the convergence of biotechnology with other technologies, like AI. However, care should be taken to not overly narrow the scope at the expense of other critical intersections.
While FAS provided a few recommendations on these topics (see Table 2), such as:
- Summary Report – December 5, 2022, Bioeconomy Policy Workshop: Measurement and Language
- Laying the Groundwork for the Bioeconomy by Sarah Carter
One of the report’s recommendations would benefit from additional stakeholder input to enhance clarity and ensure that it is actionable for Congress:
- Recommendation 8: Congress should direct the National Science Foundation (NSF) to establish a federal grant program for a national system of community biology labs that would engage Americans in informal learning. Specifically, stakeholder input on:
- What is most needed to support community biology labs?
- Should community labs be incorporated within universities or run as independent institutions?
If you have specific policy suggestions related to these topics, we encourage you to submit your ideas through the Day One Open Call at FAS.
Next Steps for the U.S. Bioeconomy
The NSCEB’s final report outlines a vision for a national biotechnology strategy aimed at securing U.S. leadership in a sector that is not only rapidly advancing but also delivering significant economic returns, outpacing even AI. While the report offers thoughtful, well-grounded recommendations that address many of the core challenges facing the U.S. biotechnology landscape, several proposals would benefit from greater specificity and refinement to make them actionable in legislative form. This moment presents a unique opportunity for stakeholders across the biotechnology ecosystem to contribute meaningfully to the development of a national bioeconomy strategy.
The U.S. bioeconomy, which encompasses biotechnology, holds enormous strategic and economic potential. Without a clear, well-implemented plan, the nation risks repeating the mistakes of past industrial shifts, such as the decline in domestic semiconductor leadership. FAS urges Congress to act on the Commission’s recommendations and leverage FAS’ additional proposals to strengthen them further (see Table 2). We also call on the scientific community to provide additional input on these recommendations to ensure they are viable and impactful. If you have actionable policy ideas on how to shape the path forward for the U.S. bioeconomy, we encourage you to submit them through the Day One Open Call. Applicants with compelling ideas will be partnered with our team at FAS to develop their idea into an implementation ready policy memo. Click here to learn more about the Day One Open Call.
FAS Bioeconomy Open Call Areas
- Recommendation 2.2d (SBIR and STTR): Congress should improve the effectiveness and reach of the Small Business Innovation Research (SBIR) and Small Business Technology Transfer (STTR) programs to support early-stage innovation.
- Which areas of biotechnology or sectors within the bioeconomy would most benefit from SBIR and STTR investment?
- How can these programs better support not only early- but also late-stage innovation?
- Recommendation 4.1c (Bio Data Generation): Congress should authorize and fund the Department of Interior to create a Sequencing Public Lands Initiative to collect new data from U.S. public lands that researchers can use to drive innovation and Recommendation 4.1d: Congress should authorize the National Science Foundation to establish a network of “cloud labs,” giving researchers state-of-the-art tools to make data generation easier.
- What type of data should be generated and what types of data generation or collection should be prioritized?
- How can we best draw on or expand existing cloud lab capabilities?
- Recommendation 4.3b (Predictable BioEngineering): Congress should initiate a grand research challenge focused on making biotechnology predictably engineerable.
- What specific grand challenge should Congress pursue and how should it be implemented?
- How should the U.S. government engage the scientific community (and others) in establishing and pursuing grand challenges for biotechnology?
- Recommendation 4.4a (Safe, Secure, Responsible): Congress must direct the executive branch to advance safe, secure, and responsible biotechnology research and innovation.
- The report calls for establishment of a body within the U.S. government for this purpose. What should this look like and how would it operate?
- Recommendation 5.2a (Domestic Bio Workforce): Congress must maximize the impact of domestic biomanufacturing workforce training programs.
- How should the government approach creating competency models for biomanufacturing training and microcredentialing?
- Which specific areas are best suited for microcredentialing efforts?
- Additional Considerations #8 (Grant Programs): Congress should direct the National Science Foundation (NSF) to establish a federal grant program for a national system of community biology labs that would engage Americans in informal learning.
- What is most needed to support community biology labs?
- Should community labs be incorporated within universities or run as independent institutions?
De-Risking the U.S. Bioeconomy by Establishing Financial Mechanisms to Drive Growth and Innovation
The bioeconomy is a pivotal economic sector driving national growth, technological innovation, and global competitiveness. However, the biotechnology innovation and biomanufacturing sector faces significant challenges, particularly in scaling technologies and overcoming long development timelines that don’t align with short-term return expectations from investors. These extended timelines and the inherent risks involved lead to funding gaps that hinder the successful commercialization of technologies and bio-based products. If obstacles like the ‘Valleys of Death, a lack of capital at crucial development junctures, that companies and technology struggle to overcome are not addressed, this could result in economic stagnation and the U.S. losing its competitive edge in the global bioeconomy.
Government programs like SBIR and STTR lessen the financial gap inherent in the U.S. bioeconomy, but existing financial mechanisms have proven insufficient to fully de-risk the sector and attract the necessary private investment. In FY24, the National Defense Authorization Act established the Office of Strategic Capital within the Department of Defense to provide financial and technical support for its 31 ‘Covered Technology Categories’, which includes biotechnology and biomanufacturing. To address the challenges associated with de-risking biotechnology and biomanufacturing within the U.S. bioeconomy, the Office of Strategic Capital within the Department of Defense should house a Bioeconomy Finance Program. This program would offer tailored financial incentives such as loans, tax credits, and volume guarantees, targeting both short-term and long-term scale-up needs in biomanufacturing and biotechnology.
By providing these essential funding mechanisms, the Bioeconomy Finance Program will reduce the risks inherent in biotechnology innovation, encouraging more private sector investment. In parallel, states and regions across the country should develop regional specific strategies, like investing in necessary infrastructure, and fostering public-private partnerships, to complement the federal government’s initiatives to de-risk the sector. Together, these coordinated efforts will create a sustainable, competitive bioeconomy that supports economic growth, and strengthens U.S. national security.
Challenge & Opportunity
The U.S. bioeconomy encompasses economic activity derived from the life sciences, particularly in biotechnology and biomanufacturing. The sector plays an important role in driving national growth and innovation. Given its broad reach across industries, impact on job creation, potential for technological advancements, and requirement for global competitiveness, the U.S. bioeconomy is a critical sector for U.S. policymakers to support. With continued development and growth, the U.S. bioeconomy promises not only economic benefits, but also strengthens national security, health outcomes, and environmental sustainability for the country.
Ongoing advancements in biotechnology, including artificial intelligence and automation, have accelerated the growth of the bioeconomy, making the sector both globally competitive and an important domestic economic sector. In 2023, the U.S. bioeconomy supported nearly 644,000 domestic jobs, contributed $210 billion to the GDP, and generated $49 billion in wages. Biomanufactured products within the bioeconomy span multiple categories (Figure 1). Growth here will drive future economic development and address societal challenges, making the bioeconomy a key priority for government investment and strategic focus.
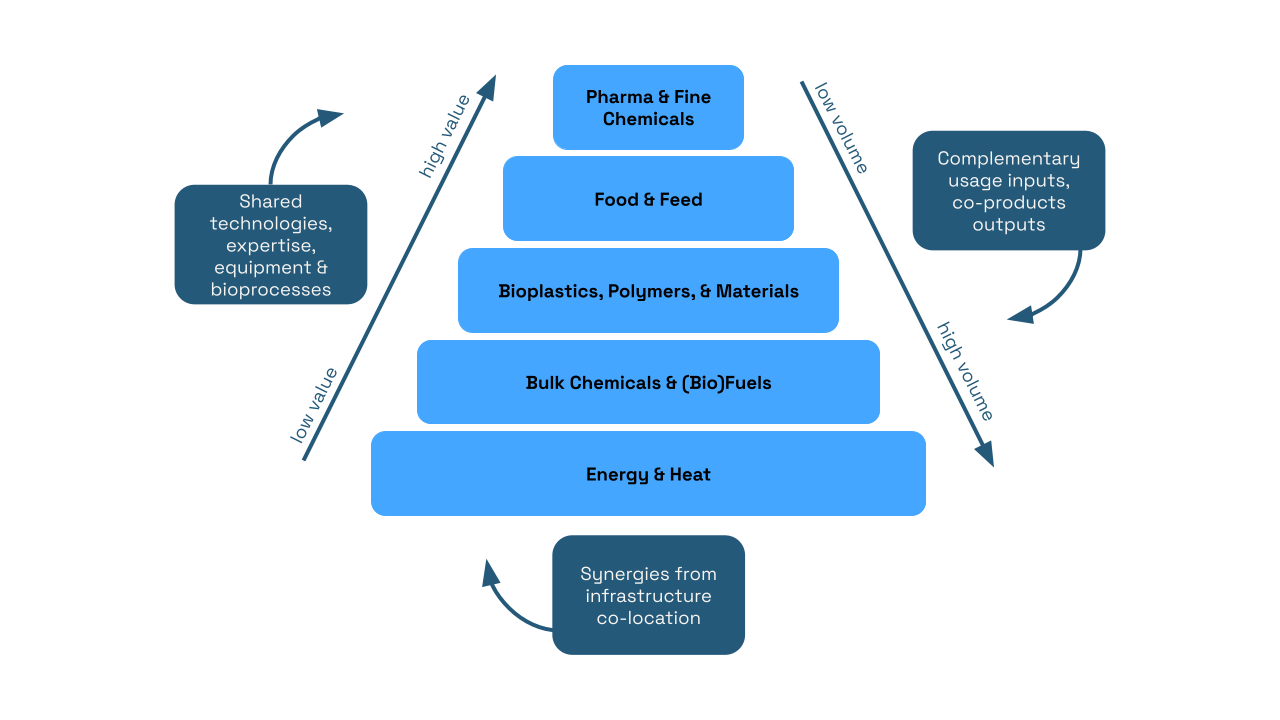
Biomanufactured products span a wide range of categories, from pharmaceuticals and chemicals, which require small volumes of biomass but yield high-value products, to energy and heat, which require larger volumes of biomass but result in lower-value products. Additionally, there are common infrastructure synergies, bioprocesses, and complementary input-output relationships that facilitate a circular bioeconomy within bioproduct manufacturing. Source: https://edepot.wur.nl/407896
An important driving force for the U.S. bioeconomy is biotechnology and biomanufacturing innovation. However, bringing biotechnologies to market requires substantial investment, capital, and most importantly, time. Unlike other technology sectors which see returns on investment within a short period of time, often, there is a misalignment between scientific and capitalistic expectations. Many biotechnology based companies rely on venture capital, a form of private equity investments, to finance their operations. However, venture capitalists (VCs) typically operate on short return on investment timelines, which may not align with the longer development cycles characteristic of the biotechnology sector (Figure 2). Additionally, the need for large-scale and the high capital expenditures (CAPEX) required for commercially profitable production, along with the low-profit margins in high-volume commodity production, create further barriers to obtaining investment. While this misalignment is not universal, it remains a challenge for many biotech startups.
The U.S. government has implemented several programs to address the financing void that often arises during the biotechnology innovation process. These include the Small Business Innovation Research (SBIR) and Small Business Technology Transfer (STTR) programs, which provide phased funding across all Technology Readiness Levels (TRLs); the DOE Loan Program Office, which offers debt financing for energy-related innovations; the DOE Office of Clean Energy Demonstrations which provides funding for demonstration-scale projects that provide proof of concept; and the newly established Office of Strategic Capital (OSC) within the DOD (as outlined in the FY24 National Defense Authorization Act), which is tasked with issuing loans and loan guarantees to stimulate private investment in critical technologies. An example is the office’s new Equipment Loan Financing through OSC’s Credit Program.
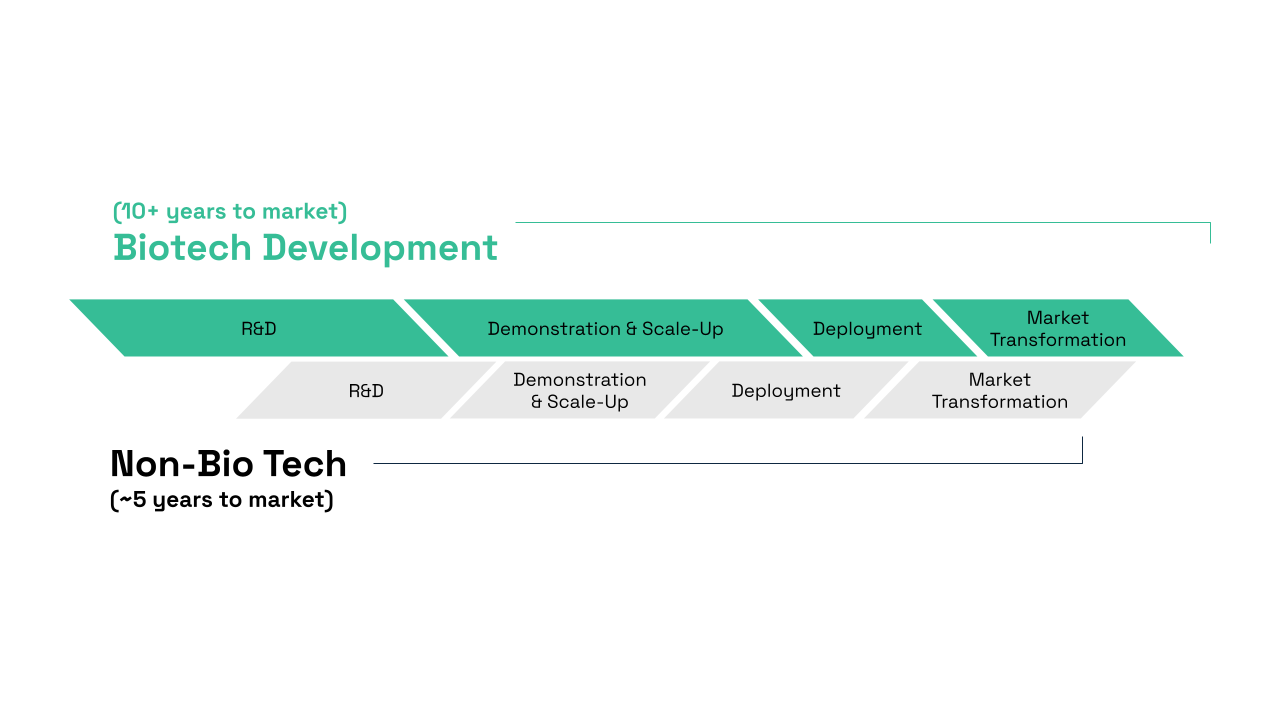
Biotechnology development timelines typically take around ~10+ years to complete and reach the market due to longer R&D and Demonstration & Scale-Up phases, while non-biotechnology development timelines are generally much shorter, averaging around ~5+ years.
While these efforts are important, they are insufficient on their own to de-risk the sector to the degree which is needed to realize the full potential of the U.S. bioeconomy. To effectively support the biotechnology innovation pipeline at critical stages, the government must explore and implement additional financial mechanisms that attract more private investment and mitigate the inherent risks associated with biotechnology innovation. Building on existing resources like the Regional Technology and Innovation Hubs, NSF Regional Innovation Engines, and Manufacturing USA Institutes, help stimulate private sector investment and are crucial for strengthening the nation’s economic competitiveness.
The newly established Office of Strategic Capital (OSC) within the DOD is well-positioned to enhance resilience in critical sectors for national security, including biotechnology and biomanufacturing, through large-scale investments. Biotechnology and biomanufacturing inherently require significant CAPEX, expenses related to the purchase, upgrade, or maintenance of physical assets. This requires substantial amounts of strategic and concessional capital to de-risk and accelerate the biomanufacturing process. By creating, implementing, and leveraging various financial incentives and resources, the Office of Strategic Capital can help build the robust infrastructure necessary for private sector engagement.
To achieve this, the U.S. government should create the Bioeconomy Finance Program (BFP) within the OSC, specifically tasked with enabling and de-risking the biotechnology and biomanufacturing sectors through financial incentives and programs. The BFP should focus on different levels of funding based on the time required to scale, addressing potential ‘Valleys of Death’ that occur during the biomanufacturing and biotechnology innovation process. These funding levels would target short-term (1-2 years) scale-up hurdles to accelerate the biotechnology and biomanufacturing process, as well as long-term (3-5 years) scale-up challenges, providing transformative funding mechanisms that could either make or break entire sectors.
In addition to the federal programs within the BFP to de-risk the sector, states and regions must also make substantial investments and collaborate with federal efforts to accelerate biomanufacturing and biotechnology ecosystems within their own areas. While the federal government can provide a top-down strategy, regional efforts are critical for supporting the sector with bottom-up strategies that complement and align with federal investments and programs, ultimately enabling a sustainable and competitive biotechnology and biomanufacturing industry regionally. To facilitate this, regions should develop and implement state-wide investment initiatives like resource analysis, infrastructure programs, and a cohesive, long-term strategy focused on public-private partnerships. The federal government can encourage these regional efforts by ensuring continued funding for biotechnology hubs and creating additional opportunities for federal investment in the future.
Plan of Action
To strengthen and increase the competitiveness of the U.S. bioeconomy, a coordinated approach is needed that combines federal leadership with state-level action. This includes establishing a dedicated Bioeconomy Finance Program within the Office of Strategic Capital to create targeted financial mechanisms, such as loan programs, tax incentives, and volume guarantees. Additionally, states must be empowered to support commercial-scale biomanufacturing and infrastructure development, leveraging tech hubs, cross-regional partnerships, and building public-private partnerships to build capacity and foster innovation nationwide.
Recommendation 1. Establish and Fund a Bioeconomy Finance Program
Congress, in the next National Defense Authorization Act, should codify the Office of Strategic Capital (OSC) within DOD and authorize the creation of a Bioeconomy Finance Program (BFP) within the OSC to provide centralized federal structure for addressing financial gaps in the bioeconomy, thereby increasing productivity and competitiveness globally. In 2024, Congress expanded the OSCs mission to offer financial and technical support to entities within its 31 ‘Covered Technology Categories,’ including biotechnology and biomanufacturing. Additionally, in order to build resilience in the sector and maintain a competitive advantage globally while also strengthening national security, these substantial expenditures should be housed within the OSC. Establishing the BFP within the OSC at the DOD would allow for a targeted focus on these critical sectors, ensuring long-term stability and resilience against political shifts.
The DOD and OSC should leverage its own funding as well as its existing partnership with the Small Business Administration to direct $1 billion to set up the BFP to create and implement initiatives aimed at de-risking the U.S. bioeconomy. The Bioeconomy Finance Program should work closely with relevant federal agencies, such as the DOE, Department of Agriculture (USDA), and the Department of Commerce (DOC), to ensure a long-term cohesive strategy for financing bioeconomy innovation and biomanufacturing capacity.
Recommendation 2. Task the Bioeconomy Finance Program with Key Initiatives
A key element of the OSC’s mission and investment strategy is to provide financial incentives and support to entities within its 31 ‘Core Technology Categories’. By having BFP design and manage these financial initiatives for the biotechnology and biomanufacturing sectors, the OSC can leverage lessons from similar programs, such as the DOE’s loan program, to address the unique needs of these critical industries, which are essential for national security and economic growth.
Currently, the OSC has launched a credit program for equipment financing. While this is a necessary first step in fulfilling the office’s mission, the program is open to all 31 ‘Core Technology Categories’, resulting in broad, dilutive funding. To accelerate the bioeconomy and reduce risks in biotechnology and biomanufacturing, it is crucial to allocate resources specifically to these sectors. Therefore, BFP should take the lead in several key financial initiatives to support the growth of the bioeconomy, including:
Loan Programs
The BFP should develop specific biotechnology enabling loan programs, in addition to the new equipment loan financing program run by the OSC. These loan programs should be modeled after those in the DOE LPO, focusing on biomanufacturing scale-up, technology transfer, and overcoming financing gaps that hinder commercialization.
Example loan programs:
- DOE Title 17 Clean Energy Financing Program
- USDA Business & Industry Loan Guarantee
- Solar Foods EU Grant/Loan
Tax Incentives
The BFP office should create tax incentives tailored to the bioeconomy, such as, transferable investment and production tax credits. For example, the 45V tax credit for production of clean hydrogen could serve as a model for similar incentives aimed at other bioproducts.
Example tax incentives:
- The Inflation Reduction Act’s transferable tax credits are the gold standard for this category.
Volume Guarantees & Procurement Support
To mitigate risks in biomanufacturing, the office should establish volume guarantees for various bioproducts, offering financial assurance to manufacturers and encouraging private sector investment. An initial assessment should be conducted to identify which bioproducts are best suited for such guarantees. Additionally, the office should explore the possibility of procurement programs to increase government demand for bio-based products, further incentivizing industry growth and innovation. This effort should be undertaken in coordination with the USDA’s BioPreferred Program to minimize redundancy and to create a cohesive procurement strategy. In addition, the BFP should look to the procurement innovations promoted by the Office of Federal Procurement Policy to find solutions for forward funding to create a functioning market.
Example Volume Guarantees & Procurement Support:
- Heavy Forging Press Infrastructure Lease Agreement
- NASA and USAF buying Fairchild semiconductors in advance of needing them, and overbought performance
- Advance Market Commitments
- Joint Venture Partnerships
- Other Transaction Authorities
Recommendation 3. Develop Pipeline Programs to Address Financial and Time Horizon Needs
Utilizing the key initiatives highlighted above, the BFP should create a two-tiered financial mechanisms pipeline and program to address both the short-term and long-term financial needs. The different financial levels could potentially include:
- Level 1 – Short Term Scale-Up (1-2 years) Programs
- Subsidized cost of electricity and other utilities (waste, wastewater treatment, natural gas, energy, etc.)
- Funding for demonstration-scale projects and early-stage engineering development. Similar to the DOEs Office of Clean Energy Demonstrations or the DODs’ Defense Industrial Base Consortium round one $1-2M engineering grants)
- Tax holidays for corporate taxes and property taxes
- Allowing accelerated depreciation to reduce tax liabilities
- Land grants or subsidies for manufacturing assets
- Fast-track permitting and site preparation to avoid long waits
- Labor and workforce subsidies
- Removal of export duties on products created in the U.S. and shipped overseas
- Level 2 – Long Term Scale-Up (3-5 years) Programs
- Large-scale transferable tax credits (either production or investment tax credits) for manufacturing. Similar to the tax credits seen in the Inflation Reduction Act for clean energy.
- Large-scale manufacturing grants
- Large-scale, low-interest manufacturing loans and loan guarantees
- Government procurement contracts or commitment for offtake, such as partial/full volume guarantees
- Government direct or indirect equity investments in biomanufacturing and biotechnology innovations
Recommendation 4. State-Level Initiatives, Infrastructure Development, and Public-Private Partnerships
While federal efforts are crucial, a bottom-up approach is needed to support biomanufacturing and the bioeconomy at the state level. The federal government can support these regional activities by providing targeted funding, policy guidance, and financial incentives that align with regional priorities, ensuring a coordinated effort toward industry growth. States should be encouraged to complement federal initiatives by developing programs that support commercial-scale biomanufacturing. Key actions include:
- State-Level Bioeconomy Resource Analysis: Each state and region should conduct their own analysis to understand the bioeconomy resources at their disposal and determine what relevant resources they would need to establish or strengthen state or regional bioeconomies. Identifying these resources will help the nation understand its true bioeconomic potential by understanding where certain biomass is contained, what facilities are available and needed to develop an economically sustainable bioeconomy, and create data to better understand the economic return on investment.
- Once the analysis is completed, States should collaborate with federal agencies like the DOE, DOC, and Economic Development Administration (EDA) to create and apply for specialized grants for commercial-scale biomanufacturing facilities based off of these analyses. Grants should prioritize non-pharmaceutical biomanufacturing to expand the scope of bioeconomy growth beyond traditional sectors.
- Utility Infrastructure Grants: Another critical area is the creation of utility infrastructure needed to support biomanufacturing, such as wastewater treatment and electricity infrastructure. States should receive targeted funding for these infrastructure projects, which are essential for scaling up production. States should take these targeted funds and establish their own granting mechanism to build necessary, regional infrastructure that is needed long-term to support the U.S. bioeconomy.
- Tech Hub Partnerships: States should leverage existing tech hubs to serve as centers for innovation in bioeconomy technologies. These hubs, which are already positioned in regions with high technological readiness, can be incentivized to partner with other regions that may not yet have robust tech ecosystems. The goal is to create a collaborative, cross-regional network that fosters knowledge-sharing and builds capacity across the country.
- Foster Public-Private Partnerships (PPP): To ensure the success and sustainability of these initiatives, states should actively foster PPPs that bring together government, industry leaders, and academic institutions. These partnerships can help align private sector investment with public goals, enhance resource sharing, and accelerate the commercialization of bioeconomy technologies. By engaging in collaborative R&D, sharing infrastructure costs, and co-developing new biotechnologies, PPPs will play a crucial role in driving innovation and economic growth in the bioeconomy sector. In addition to fostering PPPs, regions should proactively work on creating models that enable these partnerships to become self-sustaining, helping to mitigate potential financial pitfalls if partners drop out of the partnership. By not only creating PPPs, but also ensuring they become fully independent over time, the associated risks with PPPs decrease significantly.
By addressing these steps at both the federal and state levels, the U.S. can create a robust, scalable framework for financing biomanufacturing and the broader bioeconomy, supporting the transition from early-stage innovation to commercial success and ensuring long-term economic competitiveness. A good example of how this approach works is the DOE Loan Program Office, which collaborates with state energy financing institutions. This partnership has successfully supported various projects by leveraging both federal and state resources to accelerate innovation and drive economic growth. This model makes sense for biomanufacturing and biotechnology within the BFP in the OSC, as it ensures coordination between federal and state efforts, de-risks the sector, and facilitates the scaling of transformative technologies.
Conclusion
Biotechnology innovation and biomanufacturing are critical components of the U.S. bioeconomy which drives innovation, economic growth, and global competitiveness, but these sectors face significant challenges due to the misalignment of development timelines and investment cycles. The sector’s inherent risks and long development processes create funding gaps, hindering the commercialization of vital biotechnologies and products. These challenges, including the ‘Valleys of Death,’ could stifle innovation, slow down progress, and result in the U.S. losing its global leadership in biotechnology if left unaddressed.
To overcome these obstacles, a coordinated and comprehensive approach to de-risk the sector is necessary. The establishment of the Bioeconomy Finance Program (BFP) within the DOD’s Office of Strategic Capital (OSC) offers a robust solution by providing targeted financial incentives, such as loans, tax credits, and volume guarantees, designed to de-risk the sector and attract private investment. These financial mechanisms would address both short-term and long-term scale-up needs, helping to bridge funding gaps and accelerate the transition from innovation to commercialization. Furthermore, building on existing government resources, alongside fostering state-level initiatives such as infrastructure development, and public-private partnerships, will create a holistic ecosystem that supports biotechnology and biomanufacturing at every stage and will substantially de-risk the sector. By empowering regions to develop their own bioeconomy strategies and leverage local federal government programs, like the EDA Tech Hubs, the U.S. can create a sustainable, scalable framework for growth. By taking these steps, the U.S. can strengthen both its economic position but also lead the world in development of transformative biotechnologies.
BioMADE, a Manufacturing Innovation Institute sponsored by the U.S. Department of Defense, plays an important role in advancing and developing the U.S. bioeconomy. Yet, BioMADE currently funds pilot to intermediate-scale projects, rather than commercial-scale projects. This leaves a significant funding gap, creating a distinct and significant challenge for the bioeconomy.. By contrast, the BFP within OSC would complement existing efforts by specifically targeting and mitigating risks in the biotechnology and biomanufacturing pipeline that current programs do not address. Furthermore, given that BioMADE is also funded by the DOD, enhanced coordination between these programs willenable a more robust and cohesive strategy to accelerate the growth of the U.S. bioeconomy.
While Private-Public Partnerships (PPPs) are already embedded in some federal regional programs, such as the EDA Tech Hubs, not all states or regions have access to these initiatives or funding. To ensure equitable growth and fully harness the economic potential of the bioeconomy across the nation, it will be important for regions and states to actively seek additional partnerships beyond federally-driven programs. This will empower them to build their own regional bioeconomies, or microbioeconomies, by tapping into regional strengths, resources, and expertise to drive localized innovation. Moreover, federal programs like EDA Tech Hubs are often focused on advancing existing technologies, rather than fostering the development of new ones. By expanding PPPs across the biotech sector, states and regions can spur broader economic growth and innovation by holistically developing all areas of biotechnology and biomanufacturing, enhancing the overall bioeconomy.
The Emerging Reach of the Bioeconomy
On Tuesday, 4/8/25, the bipartisan National Security Commission on Emerging Biotechnology (NSCEB) released their findings on how the U.S. can support and bolster the emerging bioeconomy sector. This sector, which includes biotechnology and biomanufacturing, is increasingly important to scientists working across disciplines – and will continue to shape the economic fortunes of regions across the country.
FAS looks forward to dissecting, advancing, and advocating for the Commission’s report. FAS has been active and influential in this sector and has worked with various stakeholders and experts to advance evidence-based policy recommendations to boost the U.S. bioeconomy (more below). While the report provides an essential starting point to grow and secure our biotechnology and biomanufacturing enterprise, it will be important to advocate for the recommendations found within it, but also to add and refine recommendations to meet the ever evolving U.S. bioeconomy.
FAS is especially enthusiastic about the recommendations that emphasize prioritizing and advancing biotechnology at the national level, ensuring the U.S. maintains its innovation edge. We also strongly support the recommendations aimed at scaling biotechnologies and biomanufacturing by fostering private sector growth and leveraging various financial mechanisms. These recommendations are crucial in addressing some of the most urgent challenges facing the U.S. bioeconomy and will serve as a vital step toward establishing a dynamic and adaptable national strategy for the sector. See our policy statement for more details.
Cautious and Enthusiastic Interest
While FAS is optimistic of the impact that this report can have, it is also important for FAS to be cautious around national security issues due to our 80 year old legacy. FAS began in response to how new technologies (nuclear) could be used for war (nuclear weapons). Today we remain watchful of technologies with the potential of misuse. FAS team members involved with national security take an understandably cautionary approach. The confluence of technology and access mean that there is risk associated with bio-products, too. “This opportunity must also be balanced with a clear-eyed understanding that increasing economic competition, heating geopolitics, and advancing life sciences capabilities may change how countries and other actors view the utility of globally repugnant capabilities such as biological weapons.” said Yong-Bee Lim, Associate Director of Global Risk at the Federation of American Scientists.
The report details the importance of safeguarding the biotechnology and biomanufacturing enterprise to remain competitive at the global scale, especially with China (recall the recent semiconductor shortages). However, “it’ll be important to balance both innovation capabilities and risk as we work towards ensuring that the U.S. bioeconomy is a priority area for both the Nation and for National Security.” said Nazish Jeffery, Bioeconomy Policy Manager at the Federation of American Scientists.
Bioeconomy Presents Significant Opportunities
Still, FAS continues advocating and promoting this area with great enthusiasm. The nascent bioeconomy is more than just leading edge biology meets computational gains. There are a myriad of scientific, economic, and social benefits to be had by leveraging this new industry.
No one at FAS knows this better than Nazish Jeffery, who spearheaded efforts to understand this moment. For more than two years she has worked with biologists, technologists, policymakers and biotechnology companies to investigate how the U.S. can maintain competitiveness while distributing economic rewards equitably domestically.
FAS sponsored policy sprints, which are open calls for participants in academia and industry to submit and develop actionable policy memos that address a particular issue or sector. The goal is to infuse diverse perspectives and expertise into policy that improves lives for all Americans. Since the start of the Commission’s investigations, FAS has sponsored policy sprints on topics such as the intersection of biology and artificial intelligence, as well as a sprint soliciting ideas to grow the bioeconomy sector itself.
This emerging technology sector brought in $210 billion into the U.S. economy in 2023, and is projected to grow to $400 billion by 2030. The economic potential of the bioeconomy is significant; policymakers should promote and work in partnership with industry to continue development in distributed regions across the U.S. to invigorate innovation and enable job creation. This opportunity must also be balanced with a clear-eyed understanding that increasing economic competition, heating geopolitics, and advancing life sciences capabilities may change how countries and other actors view the utility of globally repugnant capabilities such as biological weapons.
FAS interviewed, worked with, or sought input from numerous academics, technologists, bio-industry leaders, elected representatives, and organizations, to understand the full spectrum of the value chain and push forward the best ideas.
FAS invites you to take a look at what possibilities lay ahead, presented below.
Bioeconomy Policy Sprint
The FAS Bioeconomy Sprint produced actionable policy memos to strengthen the bioeconomy in concert with outside expertise, including:
- A Matter of Trust: Helping the Bioeconomy Reach Its Full Potential with Translational Governance by Christopher Gilliespie
- BioNETWORK: The Internet of Distributed Biomanufacturing by Justin C. Sanchez
- Coordinating the U.S. Government Approach to the Bioeconomy by Sarah R. Carter
- Strengthening the U.S. Biomanufacturing Sector Through Standardization by Chris Stowers
- Accelerating Biomanufacturing and Producing Cost-Effective Amino Acids through a Grand Challenge by Allison Berke
- Project BOoST: A Biomanufacturing Test Facility Network for Bioprocess Optimization, Scaling, and Training by Ed Chung & Charles Fracchia
- Advancing the U.S. Bioindustrial Production Sector by Michael Fisher
- Accelerating Bioindustry Through Research, Innovation, and Translation by Jon Roberts
Bioeconomy x AI Policy Sprint
Artificial intelligence continues to develop exponentially; these recommendations can scale alongside AI and deliver substantial benefits:
- Develop a Screening Framework Guidance for AI-Enabled Automated Labs by Tessa Alexanian
- An Evidence-Based Approach to Identifying and Mitigating Biological Risks From AI-Enabled Biological Tools by Richard Moulange & Sophie Rose
- A Path to Self-governance of AI-Enabled Biology by Oliver Crook
- A Global Compute Cloud to Advance Safe Science and Innovation by Samuel Curtis
- Establish Collaboration Between Developers of Gene Synthesis Screening Tools and AI Tools Trained on Biological Data by Shrestha Rath
- Responsible and Secure AI in Production Agriculture by Jennifer Clarke
Additional Bioeconomy Research
Given the ongoing cost curve declines of compute, increased access to data, and growing interest in this emerging sector, FAS continues to investigate a range of related topics. Some recent work includes:
- Understanding the U.S. Bioeconomy: Agency Perspectives
- The U.S. Bioeconomy Needs Biomass, But What Is It and How Do We Use It?
- The Importance of Standards for the U.S. Bioeconomy & National Security: A Conversation with Congressman Jake Auchincloss
- Bold Goals Require Bold Funding Levels. The FY25 Requests for the U.S. Bioeconomy Fall Short
- Regulations, funding, and knowledge gaps: Challenges and opportunities in bringing agricultural biotechnology to market
- The U.S. Bioeconomy is Not Yet Sustainable. Here’s What Needs to Change.
- Implementing the Bioeconomy Executive Order: Lessons Learned and Future Considerations
- Wins, Gaps, & Looking Forward in the U.S. Bioeconomy
- “The US needs to lean into an old strength”: Maintaining Progress and Growing US Biomanufacturing
FAS will continue to work in this important area. Ongoing work related to the U.S. bioeconomy will be regularly updated here: https://fas.org/initiative/bioeconomy/
Position on National Security Commission on Emerging Biotechnology Final Report: Charting the Future of Biotechnology
The Federation of American Scientists supports the National Security Commission on Emerging Biotechnology’s Final Report and the Recommendations contained within it.
Charting the Future of Biotechnology delivers 49 recommendations to foster the growth of the biotechnology and biomanufacturing sector within the U.S. bioeconomy. Implementing the recommendations outlined in this report will strengthen the U.S. bioeconomy by establishing a unified national strategy that fosters innovation in biotechnology, ensures our continued global competitiveness, and delivers significant economic and societal benefits to the nation.
FAS is particularly excited by these recommendations:
- 1.1a: Establishment of a National Biotechnology Coordination Office
- 2.2a-d: Economic levers to promote scale-up and innovations coming to market
- 2.4a: Biotechnology infrastructure and data to be classified as “critical infrastructure”
- 3.1a: Department of Defense to consult with stakeholders to define principles for ethical use of biotechnology
- 4.3a-c: Centers for Biotechnology
- 5.1a through 5.3b: Biotechnology workforce for the future
- 6.1a-e: Strengthen global U.S. biotechnology efforts through global policy
These recommendations have the potential to address key challenges within the U.S. bioeconomy, including the lack of a coordinated strategy, commercialization barriers, workforce shortages, and supply chain vulnerabilities.
“FAS applauds the NSCEB’s deep investigation of unlocking U.S.-led biotechnology in the Fourth Industrial Revolution. We look forward to bringing FAS’s unique and effective approach of policy entrepreneurship to realize the promise of these capabilities while reducing the risks of misuse,” said Yong-Bee Lim, Associate Director of Global Risk at the Federation of American Scientists.
“The National Commission on Emerging Biotechnology report developed 50 recommendations to address the major challenges currently facing the U.S. bioeconomy: a lack of strategy and coordination across the federal government, difficulties in scaling biotechnology innovations, and the need for a trained workforce for the future. These recommendations aim to de-risk the biotechnology sector, thereby enabling private sector investment in critical biotechnology and biomanufacturing initiatives. Ultimately, these efforts will foster continued growth, secure the U.S. bioeconomy, and lead to the creation of new jobs and further economic growth.”” said Nazish Jeffery, Bioeconomy Policy Manager at the Federation of American Scientists. “It will be important to continue advocating, refining, and adding additional recommendations in order to realize the full value that this report offers.”
For more information contact Nazish Jeffery, FAS Bioeconomy Policy Manager, njeffery@fas.org.
Creating a National Exposome Project
The U.S. government should establish a public-private National Exposome Project (NEP) to generate benchmark human exposure levels for the ~80,000 chemicals to which Americans are regularly exposed. Such a project will revolutionize our ability to treat and prevent human disease. An investment of $10 billion over 20 years would fuel a new wave of scientific discovery and advancements in human health. To date, there has not been a systematic assessment of how exposures to these environmental chemicals (such as pesticides, solvents, plasticizers, medications, preservatives, flame retardants, fossil fuel exhaust, and food additives) impact human health across the lifespan and in combination with one another.
While there is emerging scientific consensus that environmental exposures play a role in most diseases, including autoimmune conditions and many of the most challenging neurodegenerative diseases and cancers, the lack of exposomic reference data restrains the ability of scientists and physicians to understand their root causes and manage them. The biomedical impact of creating a reference exposome would be no less than that of the Human Genome Project and will serve as the basis of technological advancement, the development of new medicines and advanced chemicals, and improved preventative healthcare and the clinical management of diseases.
Challenge and Opportunity
The Human Genome Project greatly advanced our understanding of the genetics of disease and helped accelerate a biotech revolution, creating an estimated $265 billion economic impact in 2019 alone. However, genetics has been unable to independently explain the root causes of the majority of diseases from which we suffer, including neurodegenerative diseases like Alzheimer’s and Parkinson’s and many types of cancer. We know exposures to chemicals and pollution are responsible for or mediate the 70–90% of disease causation not explained by genetics. However, because we lack an understanding of their underlying causal factors, many new medicines in development are more palliative than curative. If we want to truly prevent and cure the most intractable illnesses, we must uncover the complex environmental factors that contribute to their pathogenesis.
In addition to the social and economic benefits that would come from reducing our society’s disease burden, American leadership in exposomics would also strengthen the foundation of our biomedical innovation ecosystem, making the U.S. the premier partner for what is likely to be the most advanced health-related research field in this century.
Three key trends are converging to make now the best time to act: First, the costs of chemical sensors and the data and analytics infrastructure to manage them have fallen precipitously over the last two decades. Second, a few existing small scale exposomic projects offer a blueprint for how to build the NEP. Third, advancements in artificial intelligence (AI) and machine learning are making possible entirely new tools to make causal inferences from complex environmental data, which can inform research into treatments and policies to prevent diseases.
Plan of Action
To bring the National Exposome Project to life, Congress should appropriate $10 billion over 20 years to Department of Health and Human Services (HHS) to establish a National Exposomics Project Office within the Office of the HHS Secretary, whose director reports directly to the HHS Secretary. The NEP director should be given authority to establish partnerships with HHS agencies (National Institutes of Health, Centers for Disease Control, Advanced Research Projects Agency for Health, Food and Drug Administration) and other federal agencies (Environmental Protection Agency, Commerce, Defense, Homeland Security, National Science Foundation), and to fund and enter into agreements with state and local governments, academic, and private sector partners. The NEP will operate through a series of public-private cores that each are responsible for one of three pillars.
Recommendation 1. Create a reference human exposome
Through partnerships with industry, government, and academic partners, the NEP would generate comprehensive data on the body burden of chemicals and the corresponding biological responses in a representative sampling of the U.S. (>500,000 individuals). This would likely require collecting bio samples (such as blood, saliva, etc.) from participating individuals, performing advanced chemical analysis on the samples using technologies such as high- resolution mass spectrometry, and following up over the study with the participants to observe which health conditions emerge. Critically, bio samples will need to be collected repeatedly over time and bio-banked in order to ensure that the temporal aspect of exposures (such as whether someone was exposed to a particular chemical as a child or as an adult) is included in the final complete data set.
High-throughput toxicological data using microphysiological systems with human cells and tissues could also be generated on a priority list (~1000) of chemicals of concern to understand their potential harm or benefit.
These data would inform a reference standard for particular chemical exposures, which would contain the distribution of exposure levels across the population, the potential health hazards associated with a particular exposure level, and the potential combinations of exposures that would be of concern. This information could ultimately be integrated into patient electronic health records for use in clinical practice.
Recommendation 2. Develop cutting-edge data and analytical standards for exposomic analysis
The NEP would develop both a data standard for collecting and making available exposomic data to researchers, companies, and the public and advanced analytics to enable high-value causal insights to be extracted from these data to enable policymaking and scientific discovery. Importantly, the data would include both biochemical data collected directly as part of this project and in-field sensor data that is already being collected at individual, local, regional, national, and global levels by trusted third-party organizations, such as air/water quality. A key challenge in understanding the connections between a set of exposures and a disease state today is the lack of data standardization. The NEP’s investments in standardization and analytics could result in a global standard for how environmental exposure data is collected, cementing the U.S. as the global leader.
Recommendation 3. Catalyze biomedical innovation and entrepreneurship
A NEP could bolster new entrepreneurial ecosystems in advanced diagnostics, medicines, and clinical services. With access to a core reference exposome as a foundation, the ingeniousness of American entrepreneurs and scientists could result in a wellspring of innovation, creating the potential to reverse the rising incidence rates of many intractable illnesses of our modern era. One can imagine a future where exposomic tests are a part of routine physicals, informing physicians and patients exactly how to prevent certain diseases from emerging or progressing, or one where exposomic data is used to identify novel biological targets for curative therapeutics that could reverse the course of an environmentally caused disease.
The Size of the Prize
The National Exposome Project offers great potential to catalyze biomedical entrepreneurship and innovation.
First, the high-quality reference levels of exposures generated by the NEP could unlock significant opportunities in medical diagnostics. Already, great work is being done in diagnostics to understand how environmental exposures are driving diseases from autism to congenital heart defects in newborns. NEP would accelerate such work, enabling the early detection and monitoring of conditions that today have limited diagnostic approaches.
Second, a deeper understanding of exposures could lead to the faster development of new medicines. One way the NEP data set could do this would be by enabling biologists to identify novel molecular targets for medicines that might otherwise be overlooked—for example, the NEP data might reveal that certain exposures are protective and beneficial for patients with a given disease, a finding that could be more deeply examined at the molecular level to identify a novel therapeutic strategy. In addition, we know that genetics is unable to explain the hundreds of failed drug trials. Exposomics could rescue many drugs that failed testing due to environmentally related nonresponse by identifying the causative agents.
Finally, we expect that the NEP would likely result in significant advances in the physical hardware and instrumentation that is used for large-scale chemical analysis and research, and in the AI-driven computational approaches that would be necessary for the data analysis. These advancements would set the U.S. up to be the leader in exposomic sequencing and analysis, much in the same way that the Human Genome Project established the U.S. as the leader in genetic sequencing. Furthermore, these technical advances would likely be useful in many domains outside of human health where chemical analysis is useful in developing new products—such as in the agriculture, industrial chemical, and energy industries.
Conclusion
To catalyze the next generation of biomedical innovation, we need to establish a national network of exposome facilities to track human exposure levels over time, accelerate efforts to create toxicological profiles of these chemicals, develop advanced analytical models to establish causal links to human disease, and use this foundational knowledge to further the development of new medicines and policies to reduce harmful exposure. This knowledge will transform our biomedical and healthcare industries, as well as provide a path for an improved chemical industry that creates products that are safer by design. The result will be longer health spans, reductions in mortality and morbidity, and economic development associated with spurring new startups that can create new therapies, technologies, and interventions.
This action-ready policy memo is part of Day One 2025 — our effort to bring forward bold policy ideas, grounded in science and evidence, that can tackle the country’s biggest challenges and bring us closer to the prosperous, equitable and safe future that we all hope for whoever takes office in 2025 and beyond.
PLEASE NOTE (February 2025): Since publication several government websites have been taken offline. We apologize for any broken links to once accessible public data.
Anyone concerned with large government initiatives may object to the proposed budget for this project. While we acknowledge the investment needed is substantial, the upside to the public is enormous, borne out both in direct economic development benefits in new exposomic industries created as a result and in the potential demystification of a large portion of currently unexplained diseases that afflict us.
Industries responsible for manufacturing products that potentially expose populations to suspected harmful chemicals may also push back on this effort. As a response, we believe that there is abundant misinformation fueled by underpowered or poorly designed studies on chemicals, including those with more harmful reputations than data supports. A more systemic data set and a newly created industry that gives people more complete, personalized, and real-time data on exposure can not only support debunking myths but also expand the set of possible actions to mitigate exposure, taking us out of a continued cycle of finger-pointing. Indeed, such a systematic approach should reveal many positive associations with modern chemicals and health outcomes, such as preservatives reducing food-borne illness or antibiotics reducing microbial-based disease.
Governance and accountability will be critical to ensure proper stewardship of taxpayer dollars and responsible engagement with the complex set of stakeholders across the country. We therefore propose creating an external advisory committee made up of community members, industry representatives, and key opinion leaders to provide oversight over the project’s design and execution and advice and recommendations throughout all stages to the NEP director.
The first steps to realizing this vision have actually already begun. ARPA-H, the agency responsible for high-risk, high-reward research and development for health, has begun to fund some foundational exposomic work. National Institutes of Health’s All of Us program has also set a foundation for what might be possible in regards to large-scale bio-banking studies. However, to have the needed impact at scale, the NEP needs to be launched on a much bigger scale, outside of existing programs, and focus on spurring economic development and the creation of new industries.
Some of the most important factors that determine success of ambitious efforts like this are the specifics of the legislative authority, the leadership/governance structure, and how much appropriations can be made available upfront. Further, while collaboration across agencies is clear, establishing clear decision-making structure with the proper oversight is critical. This is why we believe creating a dedicated program office, with a clear leader who reports directly to a member of the cabinet, endowed with the necessary authorities including Other Transactions Authority, is key to success.
Regulations, funding, and knowledge gaps: Challenges and opportunities in bringing agricultural biotechnology to market
Innovations in agriculture will play an increasingly important role in America’s quest to ensure resilient and sustainable production of food, medicine, and bioenergy products. Biotechnology, spurred by advances such as cheap sequencing, offers a realm of possibilities for novel agricultural inputs, such as more targeted pesticides that are less toxic and less likely to cause tolerance, less carbon-intensive alternatives to fertilizers, and more climate-resilient crop varieties.
However, research and development of new agricultural biotech products can be expensive and time-consuming, due to the large physical scale and long timelines of field trials. At the same time, federal funding for agriculture research has historically paled in comparison to funding for defense, energy, and human health. For example, in 2022, the NIH’s R&D budget was more than 16 times that of the USDA’s.
The Biden administration has demonstrated its recognition of the need to accelerate research and development in agricultural biotechnology, featuring it prominently in 2022’s Executive Order on Advancing Biotechnology and Biomanufacturing Innovation for a Sustainable, Safe, and Secure American Bioeconomy. Additionally, a bill to expand authorization of funding for a moonshot USDA research grant program, AgARDA (Agriculture Advanced Research and Development Authority), has broad bipartisan support. At the same time, there has been a commensurate increase in private funding.
While this multi-front surge in enthusiasm and investment is welcome, many challenges remain in translating money, ideas, and laboratory results to the field and the market, including communication between the various stakeholders in agricultural biotechnology R&D. To better understand industry priorities and potential barriers to progress, we spoke to members of the executive team of Fall Line Capital (FLC), a venture capital (VC) and private equity firm that invests in food/agriculture startups. Fall Line’s investments include new biopesticides (Greenlight, Micropep), functional microbes (Pluton, Wild Microbes), and new equipment (Guardian Agriculture, Rantizo, LUMO), in addition to managing a farmland portfolio. As lifelong farmers as well as agriculture technology (agtech) investors, Clay Mitchell and Scott Day offer a multifaceted perspective on the current landscape.
We then outline actions for government actors that can address the challenges identified in our interview, in three key areas: regulatory oversight, federal R&D funding, and bioliteracy.
Q: What can the U.S. government do to provide a supportive landscape for new agricultural biotechnology?
Fall Line Capital: I think the biggest hurdles are regulatory. If the government wants to be truly supportive and innovative, it should be working to revamp the convoluted regulatory environment. The current system wasn’t designed to handle all the new technology being developed with novel mechanisms of action, so hurdles to creating and commercializing stifle innovation even more than they did in the past.
Looking at new technology like RNA– and micropeptide-based pesticides, it’s been a difficult process to get those products registered, even though they should be embraced: compared to conventional pesticides, they have the potential to be highly specific to the target organism and minimally toxic to non-target organisms. During GreenLight’s discussions with the EPA to register their RNAi biopesticide for tackling invasive potato beetles, the EPA seemed to understand that this sort of technology is the future, but movement through the registration approval process was slow nevertheless; the application sat there for five 5 years. There were dozens of other biological pesticide product applications, and the EPA had to give every application the same level of scrutiny, even if many were obviously ineffective. There’s pressure to register more biological products as a prominent alternative to traditional chemical products, despite generally low efficacy. This clutters up the process, and the EPA was already short-staffed after extensive attrition during COVID.
A substantial amount of the innovation is coming from small companies like Greenlight that don’t have the resources (which many of the large incumbent ag companies have) to navigate the current registration programs and protocols, which are spread across multiple agencies involved in regulating biotechnology: the USDA, EPA, and FDA. There needs to be a new concierge resource beyond what the Unified Website for Biotechnology Regulation currently provides, that could direct you to the right office, the right registration process, as well as appropriate funding opportunities and legal resources.
Q: What do you think are currently the most pressing challenges in agriculture?
FLC: Pest resistance continues to be a very serious concern in agriculture, so new and effective control measures need to be continually developed for all pests: weeds, insects, disease. There are two major pests of concern for my own farm in Western Canada. First, herbicide-resistant kochia weed, which has become a huge problem in the last five years all across the world. No one’s sure how exactly it spread so quickly. Second, flea beetles are decimating cruciferous crops. RNAi-based insecticides could be very effective here, if we can achieve sufficient persistence of the insecticide and avoid impacting non-target species.
In terms of challenges to agriculture-based businesses, there’s a lack of funding right now for getting tech to market. Funding for agtech from VC firms fell last year, as it did for most forms of tech. This was following a very strong period of agtech funding for the previous two years, during which we saw over-investment in several sectors, such as alternative meat and indoor farming. At the same time, agtech companies typically have long timelines to product launch and need more funding than just one VC can provide. Right now, many companies who come to Fall Line for money are just looking for short-term “bridge funding” so they can make payroll and buy time until they can demonstrate enough progress to raise a successful “series” round with good valuation and favorable investment terms. And no one is going public right now.
Q: What are common knowledge gaps for agtech startups regarding farmers’ needs?
FLC: Agtech startups are often centered around a great idea or technology that’s looking for a problem to solve — but it’s hard for a specific technology to meet the needs of a variable problem. Farmers’ needs and priorities (e.g. pests, nutrients, etc.) are incredibly diverse, varying dramatically by crop and location, even from one field to the next on the same farm, or even within the same field. Today, it is very hard to get an accurate understanding of what is needed or desired at the farm level because there is no easy way to connect with growers on a broad scale. Farm papers have diminished in popularity just like mainstream papers, radio has diminished as well. Unless you have the email address or cell phone number of a farmer, it is hard to connect directly with them now, and most farmers don’t like doing surveys of any type anyway — and those that do aren’t that representative of the industry. I think this is why most types of polls are becoming less accurate as it is increasingly more difficult to get a representative sample of opinions.
Farmers can be hesitant to adopt new technologies, since the risk can be high. And once they’ve been burned once by a product that failed to work as advertised, they’re unlikely to be willing to trust that company, or even that type of product, in the future. For example, last year, North Dakota State University scientists coordinated a large-scale field trial where it showed in a large field trial that most new biological products aimed at improving nitrogen-fixation in non-legume crops were ineffective at increasing yield. In general, the efficacy of biologicals can vary greatly depending on the exact field conditions, making it hard to reliably achieve the advertised result. There’s a huge jump from greenhouse results to field trials, another huge jump from field trials to commercial fields. But when a product’s value is obvious, farmers actually embrace new technology very quickly: both GMO crops and GPS achieved widespread adoption in a very short period of time.
Finally, technology developers should keep in mind that problems can be solved by old or simple technology. When people think about controlled-environment farming, their minds jump to fancy things like vertical farming — but with irrigation and mulch films, you’re 90% of the way there. Simply by adding a mulch film to heat the soil, farmers can greatly extend the growing season in northern climates by a month. This approach allowed us us to substantially increase the yield from our corn fields in Wisconsin.
This conversation illustrates a clear need for change in three key areas:
Federal funding for agricultural R&D
Given the unreliability of private market funding for agricultural biotechnology R&D, which often entails long turnaround times and low margins relative to traditional tech companies, substantial federal funding through research programs such as AgARDA is vital for accelerating R&D. AgARDA, based on the ARPA Advanced Research Projects Agency model, would allow the USDA to support the development of transformative technologies for focus areas of its choosing. However, despite its popularity, AgARDA, which was first authorized in the 2018 Farm Bill for $50 million annually for FY2019-2023, only received $2m in that timeframe. The USDA requested $5m for AgARDA in FY2022 and again in FY2023; it only received $1m each year. By contrast, ARPA-H, the human health equivalent, was authorized in FY2022 and immediately received its full $1 billion authorization, followed by $1.5b in FY2023.
The USDA has published an implementation framework for AgARDA. Unfortunately, misalignment between USDA and Congress appears to be preventing AgARDA from being fully funded to its authorized levels. Members of the Congressional agriculture committees want the USDA to show that it has made progress with the $2m it has received before they allocate additional funding, namely the appointment of a dedicated director and initiation of a pilot program with calls for grant proposals. However, the USDA has deemed the $2m insufficient to support long-term staff or a formal grant program, especially since the appropriations require annual renewal. The current impasse means that no AgARDA projects have been rolled out, despite the pressing nature of the research priorities identified by the USDA.
The following steps should be taken for AgARDA to achieve its full potential:
- USDA should produce a formal report to Congress of how it utilized the $2m thus far allocated.
- Congress should ensure that the bill to expand AgARDA authorization from $50m to $100m is passed as part of the upcoming Farm Bill.
- USDA should request, and Congress should approve, funding up to its full authorization for AgARDA in its FY2026 budget. Additionally, the appropriations should be granted on a three-year basis, like ARPA-H’s appropriations, to permit greater runway.
Regulatory oversight
The U.S. regulatory system for biotechnology needs to be a) expanded, with funding for a larger agency staff to process applications quickly; b) updated, to be flexible such that it can accommodate new-to-market technologies; and c) coordinated, to streamline approval processes.
The National Security Commission on Emerging Biotechnology (NSCEB) addresses these unmet needs in its interim report. First, NSCEB is “considering options to facilitate higher staffing levels”; this should be made a priority.
Second, concerning regulatory oversight, NSCEB identified three potential paths for improvement:
- discrete changes to individual statutes to reduce redundancies and gaps in biotechnology oversight;
- a single, unified regulatory process to assess any novel risks associated with biotechnology products relative to their conventional counterparts; and
- a hybrid approach that legislatively mandates coordination while facilitating individual agency review and risk assessment.
Of these, the hybrid approach would likely provide the greatest flexibility. In contrast, discrete changes to individual statutes will likely involve slow, piecemeal changes that can easily become outdated again. While a unified regulatory process may be more streamlined, the report’s phrasing creates a sharp binary delineation between biotech and conventional that does not reflect reality. Such a delineation could engender a lot of wasted time debating biotech versus conventional classification for a given product.
Finally, to address intra- and interagency coordination, the NSCEB presented two Farm Bill amendments that deserve Congressional support: the Biotechnology Oversight Coordination Act and the Agriculture Biotechnology Coordination Act.
Bioliteracy and agricultural education
Market demand and regulations are informed by consumer perceptions, which then impact R&D decisions. For example, fear of consumer and regulatory backlash can dissuade companies from investing in new genetic engineering technology for developing new plant varieties, despite their potential to improve agricultural sustainability. Increased bioliteracy across the American public would help consumers, businesses, and policymakers alike better understand new biotechnologies and engage with the burgeoning bioeconomy. This is a need that the NSCEB has also highlighted. At the K-12 level, improvements could comprise updating science curriculums to include contemporary topics like gene editing, as well as amending civics curriculums to better explain the modern functions of regulatory agencies. In addition, agricultural education can be embedded into biology and earth science curriculums to reconnect the public at large with the realities faced by producers. Similar to computer science literacy improvements through standard setting and funding, bioliteracy can be improved through state-level education initiatives.
The Federation of American Scientists values diversity of thought and believes that a range of perspectives — informed by evidence — is essential for discourse on scientific and societal issues. Contributors allow us to foster a broader and more inclusive conversation. We encourage constructive discussion around the topics we care about.
BioNETWORK: The Internet of Distributed Biomanufacturing
Summary
The future of United States industrial growth resides in the establishment of biotechnology as a new pillar of industrial domestic manufacturing, thus enabling delivery of robust supply chains and revolutionary products such as materials, pharmaceuticals, food, energy. Traditional centralized manufacturing of the past is brittle, prone to disruption, and unable to deliver new products that leverage unique attributes of biology. Today, there exists the opportunity to develop the science, infrastructure, and workforce to establish the BioNETWORK to advance domestic distributed biomanufacturing, strengthen U.S.-based supply chain intermediaries, provide workforce development for underserved communities, and achieve our own global independence and viability in biomanufacturing. Implementing the BioNETWORK to create an end-to-end distributed biomanufacturing platform will fulfill the Executive Order on Advancing Biotechnology and Biomanufacturing Innovation and White House Office of Science and Technology Policy (OSTP) Bold Goals for U.S. Biotechnology and Biomanufacturing.
Challenge and Opportunity
Biotechnology harnesses the power of biology to create new services and products, and the economic activity derived from biotechnology and biomanufacturing is referred to as the bioeconomy. Today, biomanufacturing and most other traditional non-biomanufacturing is centralized. Traditional manufacturing is brittle, does not enhance national economic impact or best use national raw materials/resources, and does not maximize innovation enabled by the unique workforce distributed across the United States. Moreover, in this era of supply chain disruptions due to international competition, climate change, and pandemic-sized threats (both known and unknown), centralized approaches that constitute a single point of attack/failure and necessarily restricted, localized economic impact are themselves a huge risk. While federal government support for biotechnology has increased with recent executive orders and policy papers, the overarching concepts are broad, do not provide actionable steps for the private sector to respond to, and do not provide the proper organization and goals that would drive outcomes of real manufacturing, resulting in processes or products that directly impact consumers. A new program must be developed with clear milestones and deliverables to address the main challenges of biomanufacturing. Centralized biomanufacturing is less secure and does not deliver on the full potential of biotechnology because it is:
- Reliant on a narrow set of feedstocks and reagents that are not local, introducing supply chain vulnerabilities that can halt bioproduction in its earliest steps of manufacturing.
- Inflexible for determining the most effective, stable, scalable, and safe methods of biomanufacturing needed for multiple products in large facilities.
- Serial in scheduling, which introduces large delays in production and limits capacity and product diversity.
- Bespoke and not easily replicated when it comes to selection and design of microbial strains, cell free systems, and sequences of known function outside of the facility that made them. Scale-up and reproducibility of biomanufacturing products are limited.
- Creating waste streams because circular economies are not leveraged.
- Vulnerable to personnel shortages due to shifting economic, health, or other circumstances related to undertraining of a biotechnology specialized workforce.
Single point failures in centralized manufacturing are a root cause of product disruptions and are highlighted by current events. The COVID-19 pandemic revealed that point failures in the workforce or raw materials created disruptions in the centralized manufacturing, and availability of hand sanitizers, rubber gloves, masks, basic medicines, and active pharmaceutical ingredients impacted every American. International conflict with China and other adversarial countries has also created vulnerabilities in the sole source access to rare earth metals used in electronics, batteries, and displays, driving the need for alternate options for manufacturing that do not rely on single points of supply. To offset this situation, the United States has access to workforce, raw materials, and waste streams geographically distributed across the country that can be harnessed by biomanufacturing to produce both health and industrial products needed by U.S. consumers. However, currently there are only limited distributed manufacturing infrastructure development efforts to locally process those raw materials, leaving societal, economic, and unrealized national security risks on the table. Nation-scale parallel production in multiple facilities is needed to robustly create products to meet consumer demand in health, industrial, energy, and food markets.
The BioNETWORK inverts the problem of a traditional centralized biomanufacturing facility and expertise paradigm by delivering a decentralized, resilient network enabling members to rapidly access manufacturing facilities, expertise, and data repositories, as needed and wherever they reside within the system, by integrating the substantial existing U.S. bioindustrial capabilities and resources to maximize nationwide outcomes. The BioNETWORK should be constructed as an aggregate of industrial, academic, financial, and nonprofit entities, organized in six regionally-aligned nodes (see figure below for notional regional distribution) of biomanufacturing infrastructure that together form a hub network that cultivates collaboration, rapid technology advances, and workforce development in underserved communities. The BioNETWORK’s fundamental design and construction aligns with the need for new regional technology development initiatives that expand the geographical distribution of innovative activity in the U.S., as stated in the CHIPS and Science Act. The BioNETWORK acts as the physical and information layer of manufacturing innovation, generating market forces, and leveraging ubiquitous data capture and feedback loops to accelerate innovation and scale-up necessary for full-scale production of novel biomaterials, polymers, small molecules, or microbes themselves. As a secure network, BioNETWORK serves as the physical and virtual backbone of the constituent biomanufacturing entities and their customers, providing unified, distributed manufacturing facilities, digital infrastructure to securely and efficiently exchange information/datasets, and enabling automated process development. Together the nodes function in an integrated way to adaptively solve biotechnology infrastructure challenges as well as load balancing supply chain constraints in real-time depending on the need. This includes automated infrastructure provisioning of small, medium, or large biomanufacturing facilities, supply of regional raw materials, customization of process flow across the network, allocation of labor, and optimization of the economic effectiveness. The BioNETWORK also supports the implementation of a national, multi-tenant cloud lab and enables a systematic assessment of supply chain capabilities/vulnerabilities for biomanufacturing.
As a secure network, BioNETWORK serves as the physical and virtual backbone of the constituent biomanufacturing entities and their customers, providing unified, distributed manufacturing facilities, digital infrastructure to securely and efficiently exchange information/datasets, and enabling automated process development.
Plan of Action
Congress should appropriate funding for an interagency coordination office co-chaired by the OSTP and the Department of Commerce (DOC) and provide $500 million to the DOC, Department of Energy (DOE), and Department of Defense (DOD) to initiate the BioNETWORK and use its structure to fulfill economic goals and create industrial growth opportunities within its three themes:
- Provide alternative supply chain pathways via biotechnologies and biomanufacturing to promote economic security. Leverage BioNETWORK R&D opportunities to develop innovative biomanufacturing pathways that could address supply chain bottlenecks for critical drugs, chemicals, and other materials.
- Explore distributed biomanufacturing innovation to enhance supply chain resilience. Leverage BioNETWORK R&D efforts to advance flexible and adaptive biomanufacturing platforms to mitigate the effects of supply chain disruptions.
- Address standards and data infrastructure to support biotechnology and biomanufacturing commercialization and trade. Leverage BioNETWORK R&D needed to enable data interoperability across the network to enable scale-up and increase global competitiveness.
To achieve these goals, the policy Plan of Action includes the following steps:
1. Congress should appropriate $10 million to establish an interagency coordination office within OSTP that is co-chaired by the DOC. This fulfills the White House Executive Order and CHIPs and Science mandates for better interagency coordination among the DOE, DOC, DOD, National Institute of Standards and Technology (NIST), and the National Science Foundation (NSF).
2. Congress should then appropriate $500 million to DOC and DOE to fund a biomanufacturing moonshot that includes creating the pilot network of three nodes to form the BioNETWORK in regions of the U.S. within six months of receiving funding. This funding should be managed by the interagency coordination office in collaboration with a not-for-profit organization whose mission is to build, deploy, and manage the BioNETWORK together with the federal entities. The role of the not-for-profit is to ensure that a trusted, unbiased partner (not influenced by outside entities) is involved, such that the interests of the taxpayer, U.S. government, and commercial sectors are all represented in the most beneficial way possible. The mission should include education, workforce development, safety/security, and sustainment as core principles, such that the BioNETWORK can stand alone once established. The new work to build the network should also synergize with the foundational science of the NSF and the national security focus of DOD biotechnology programs.
3. Continued investment of an additional $500 million should be appropriated by Congress to create economic incentives to sustain and transition the BioNETWORK from public funding to full commercial operation. This step requires evaluation of concrete go/no-go milestones and deliverables to ensure on-time, on-budget operations have been met. The interagency coordination office should work with DOC, DOE, DOD, and other agencies to leverage these incentives and create other opportunities to promote the BioNETWORK so that it does not require public funding to keep itself sustainable and can obtain private funding.
Create a Pilot Network of Three Nodes
To accelerate beyond current biomanufacturing programs and efforts, the first three nodes of the BioNETWORK should be constructed in three new disparate geographic regions (i.e., East, Midwest, West, or other locations with relevant feedstocks, workforce, or component infrastructure) to show the networking capabilities for distributed manufacturing. The scale of funding required to design, construct, and deploy the first three nodes is $500 million. The initiation and construction of the BioNETWORK should commence within six months. The DOE should lead the initiation and deployment of the technical construction of the BioNETWORK through Theme 2 of their Biomanufacturing goals, which “seeks alternative processes to produce chemicals and materials from renewable biomass and intermediate feedstocks by developing low-carbon-intensity product pathways and promoting a circular economy for materials.” Each node should create regional partnerships that have four entities (a physical manufacturing facility, a cell programming entity, an academic research and development entity, and a workforce/resource entity). All four entities will contain both physical facilities such as industrial fermentation and wet lab space, as well as the workforce needed to run them. On top of the pilot nodes, a science and technology/engineering integrator of the system should be identified to coordinate the effort and lead security/safety efforts for the physical network. Construction of the initial BioNETWORK should be completed within two years.
Achievement of the BioNETWORK goals requires the design plan to:
- Leverage and use regional feedstocks and reagents across the U.S. as inputs to bioproduction to create robustness in the earliest steps of manufacturing.
- Automate the integrated use of small, intermediate, and large-scale biomanufacturing facilities so that they are effective, stable, scalable, and safe for biomanufacturing demand.
- Parallelize scheduling of infrastructure and resources to minimize delays in production and maximize capacity and product diversity.
- Incorporate methods for replication when it comes to selection and design of microbial strains, cell free systems, and sequences of known function.
- Reuse waste streams to create circular economies.
- Include infrastructure biomanufacturing standards from NIST.
The BioNETWORK construction milestones should fulfill the White House OSTP bold goals through new capabilities delivered via distributed manufacturing infrastructure:
- Networked data for distributed biomanufacturing—“establishing a Data Initiative to ensure that high-quality, wide-ranging, easily accessible, and secure biological data sets can drive breakthroughs for the U.S. bioeconomy.”
- Domestic distributed biomanufacturing infrastructure—“expanding domestic capacity to manufacture all the biotechnology products we invent in the United States and support a resilient supply chain.”
- Local hubs for workforce development—“growing training and education opportunities for the biotechnology and biomanufacturing workforce of the future.”
Full Network: Plan for Sustainability
Congress and executive branch agencies establish economic incentives for commercial entities, state/local governments, and consumers of bioindustrial manufacturing products to create commercialization pathways that enhance local economies while also supporting the national network. These include tax credits, tax breaks, low interest loans, and underwritten loans as a starting point. To facilitate tech transition, unique lab-to-market mechanisms and proven tools to address market failure and applied technologies gaps should be used in conjunction with those in the Inflation Reduction Act. This includes prize and challenge competitions, market shaping procurement or loan programs, and streamlined funding of open, cross-disciplinary research, and funding at the state and local levels.
A new public-private partnership could coordinate across multiple efforts to ensure they drive toward rapid technology deployment and integration. This includes implementing a convertible debt plan that rewards BioNETWORK members with equity after reaching key milestones, providing an opportunity for discounted buyout by other investors during rounds of funding, and working with the federal government to design market-shaping mechanisms such as advance market commitments to guarantee purchase of a bioproduction company’s spec-meeting product.
Additionally, the BioNETWORK should be required to expand the repertoire of domestic renewable raw materials into a suite of high-demand, industry-ready products as prescribed in the DOC’s goals in biomanufacturing. This will ensure all regions have support for commercial goods and can automatically assess domestic supply chain capabilities and vulnerabilities, and are provided compensatory remediation on demand. The full BioNETWORK consists of six nodes—aligned to each of the major geographic regions and/or EDA regions in the United States—which have unique raw materials, workforce, infrastructure, and consumption of products that contribute to supporting the overall network functionality. The full BioNETWORK should be active within five years of project initiation and be evaluated against phased milestones throughout.
Conclusion
Networked solutions are resilient and enduring. A single factory is at risk of transfer to foreign ownership, closure, or obsolescence. The BioNETWORK creates connectivity among distributed biomanufacturing physical infrastructure to form a network with a robust domestic value chain. Today’s biomanufacturing investments suffer from the need to vertically integrate due to lack of flexible capacity across the value chain, which raises capital requirements and overall risk. The BioNETWORK drives horizontal integration through the network nodes via new infrastructure, connecting physical infrastructure of the nodes within the system. The result is a multi-sided marketplace for biotechnology innovation, products, and commercialization.
The federal government should initiate a new program and select performers within the next six months to begin the research, development, and construction of the first three nodes of the BioNETWORK. Taking action to establish the BioNETWORK ensures that the United States has the necessary physical and virtual infrastructure to grow the bioeconomy and its international leadership in biotechnology. The BioNETWORK creates new job opportunities for people across the country where training in biotechnology expands the skill sets of people with broad-spectrum applicability from trades to advanced degrees. The BioNETWORK drives circular economies where raw materials from rural and urban centers enter the network and are transformed into high-value products such as advanced materials, pharmaceuticals, food, and energy. The BioNETWORK protects U.S. supply chain resiliency through distributed manufacturing and links regional development into a national capability to establish biomanufacturing as a pillar of economic and technological growth for today and into the 22nd century.
Establishment of the BioNETWORK scales, connects, and networks the impact of a hub and tailors it to the needs of bioindustrial manufacturing, which requires regional feedstocks and integration of small-, intermediate-, and large-scale industrial fermentation facilities scattered across the United States to form an end-to-end distributed biomanufacturing platform. Similar to the goals of the EDA hub program, the BioNETWORK will accelerate regional economic activity, workforce development, and re-establishment of domestic manufacturing. Leveraging activity of the EDA and NSF Biofoundries program is an opportunity for coordination across the interagency.
Retrofitting existing small-, intermediate-, and large-scale biomanufacturing facilities/plants is necessary to construct the connected BioNETWORK. This includes new/modified fermentation equipment, scale-up and purification hardware, software/communications for networking, transportation, load-balancing, and security infrastructure.
Clear, measurable intermediate milestones and deliverables are required to ensure that the BioNETWORK is on track. Every three months, key performance metrics and indicators should be used to demonstrate technical functionality. Planned economic and workforce targets should be established every year and tracked for performance. Adjustments to the technical and business plans should be implemented if needed to ensure the overarching goals are achieved.
A major outcome of the BioNETWORK program is that biomanufacturing in the United States becomes on par with the other traditional pillars of manufacturing such as chemicals, food, and electronics. Workforce retraining to support this industry leads to new high-paying jobs as well as new consumer product sectors and markets with new avenues for economic growth. Failure to deploy the BioNETWORK leaves the United States vulnerable to supply chain disruption, little to no growth in manufacturing, and out competition by China and other peer nations that are investing in and growing biotechnology.
Secondary milestones include key performance indicators, including increased capacity, decrease in production time, robustness (more up time vs. down time), cheaper costs, better use of regional raw materials, etc.
“The US needs to lean into an old strength”: Maintaining Progress and Growing US Biomanufacturing
The U.S. bioeconomy has been surging forward, charged by the Presidential Executive Order 14081 and the CHIPS and Science Act. However, there are many difficult challenges that lay ahead for the U.S. bioeconomy, including for U.S. biomanufacturing capabilities. U.S. biomanufacturing has been grappling with issues in fermentation capacity including challenges related to scale-up, inconsistent supply chains, and downstream processing. While the U.S. government works on shoring up these roadblocks, it will be important to bring industry perspectives into the conversation to craft solutions that not only addresses the current set of issues but looks to mitigate challenges that may arise in the future.
To get a better understanding of industry perspectives on the U.S. bioeconomy and the U.S. biomanufacturing sector, the Federation of American Scientists interviewed Dr. Sarah Richardson, the CEO of MicroByre. MicroByre is a climate-focused biotech startup that specializes in providing specialized bacteria based on the specific fermentation needs of its clients. Dr. Richardson received her B.S. in biology from the University of Maryland in 2004 and a Ph.D. in human genetics and molecular biology from Johns Hopkins University School of Medicine in 2011. Her extensive training in computational and molecular biology has given her a unique perspective regarding emerging technologies enabled by synthetic biology.
FAS: The U.S. Government is focused on increasing fermentation capacity, including scale-up, and creating a resilient supply chain. In your opinion, are there specific areas in the supply chain and in scale-up that need more attention?
Dr. Sarah Richardson: The pandemic had such an impact on supply chains that everyone is reevaluating the centralization of critical manufacturing. The United States got the CHIPS and Science Act to invest in domestic semiconductor manufacturing. The voting public realized that almost every need they had required circuits. Shortages in pharmaceuticals are slowly raising awareness of chemical and biomedical manufacturing vulnerabilities as well. The public has even less insight into vulnerabilities in industrial biomanufacturing, so it is important that our elected officials are proactive with things like Executive Order 14081.
When we talk about supply chains we usually mean the sourcing and transfer of raw, intermediate, and finished materials — the flow of goods. We achieve robustness by having alternative suppliers, stockpiles, and exacting resource management. For biomanufacturing, an oft raised supply chain concern is feedstock. I can and will expound on this, but securing a supply of corn sugar is not the right long-term play here. Shoring up corn sugar supplies will not have a meaningful impact on industrial biomanufacturing and should be prioritized in that light.
Biomanufacturing efforts are different from the long standing production of consumer goods in that they are heavily tied to a scientific vendor market. As we scale to production, part of our supply chain is a lot of sterile plastic disposable consumables. We compete with biomedical sectors for those, for personal protective equipment, and for other appliances. This supply chain issue squeezed not just biomanufacturing, but scientific research in general.
We need something that isn’t always thought of as part of the supply chain: specialized infrastructural hardware. This may not be manufactured domestically. Access to scale up fermentation vessels is already squeezed. The other problem is that no matter where you build them, these vessels are designed for the deployment of canonical feedstocks and yeasts. Addressing the manufacturing locale would offer us the chance to innovate in vessel and process design and support the kinds of novel fermentations on alternate feedstocks that are needed to advance industrial biomanufacturing. There are righteous calls for the construction of new pilot plants. We should make sure that we take the opportunity to build for the right future.
One of the indisputable strengths of biomanufacturing is the potential for decentralization! Look at microbrewing: fermentation can happen anywhere without country-spanning feedstock pipelines. As we onboard overlooked feedstocks, it may only be practical to leverage them if some fermentation happens locally. As we look at supply chains and scale up we should model what that might look like for manufacturing, feedstock supply chains, and downstream processing. Not just at a national level, but at regional and local scales as well.
There are a lot of immediate policy needs for the bioeconomy, many of which are outlined in Executive Order 14081. How should these immediate needs be balanced with long-term needs? Is there a trade-off?
Counterintuitively, the most immediate needs will have the most distant payoffs! The tradeoff is that we can’t have every single detail nailed down before work begins. We will have to build tactically for strategic flexibility. Climate change and manufacturing robustness are life or death problems. We need to be open to more creative solutions in funding methods, timeline expectations; in who comes to the table, in who around the table is given the power to affect change, and in messaging! The comfortable, familiar, traditional modes of action and funding have failed to accelerate our response to this crisis.
We have to get started on regulation yesterday, because the only thing that moves slower than technology is policy. We need to agree on meaningful, aggressive, and potentially unflattering metrics to measure progress and compliance. We need to define our terms clearly: what is “bio-based,” does it not have petroleum in it at all? What does “plant-based” mean? What percentage of a product has to be renewable to be labeled so? If it comes from renewable sources but its end-of-life is not circularizable, can we still call it “green”?
We need incentives for innovation and development that do not entrench a comfortable but unproductive status quo. We need to offer stability to innovators by looking ahead and proactively incubating the standards and regulations that will support safety, security, and intellectual property protection. We should evaluate existing standards and practices for inflexibility: if they only support the current technology and a tradition that has failed to deliver change, they will continue to deliver nothing new as a solution.
We need to get on good footing with workforce development, as well. A truly multidisciplinary effort is critical and will take a while to pull off; it takes at least a decade to turn a high school student into a scientist. I only know of one national graduate fellowship that actually requires awardees to train seriously in more than one discipline. Siloing is a major problem in higher education and therefore in biomanufacturing. What passes for “multidisciplinary” is frequently “I am a computer scientist who is not rude to biologists” or “our company has both a chemical division and an AI division.” A cross-discipline “bilingual” workforce is absolutely critical to reuniting the skill sets needed to advance the bioeconomy. Organizations like BioMADE with serious commitments to developing a biomanufacturing workforce cannot effectively address the educational pipeline without significantly more support.
MicroByre is working to advance alternatives to substrates currently favored by the bioeconomy.
When we emphasize the collection of data — which data are we talking about? Is the data we have collected already a useful jumping off point for what comes next? Are the models relevant for foreseeable changes in technology, regulation, and deployment? For some of it, absolutely not. As every responsible machine learning expert can tell you, data is not something you want to skimp or cheap out on collecting or curating. We have to be deliberate about what we collect, and why. Biases cannot all be avoided, but we have to take a beat to evaluate whether extant models, architecture, and sources are relevant, useful, or adaptable. A data model is as subject to a sunk cost fallacy as anything else. There will be pressure to leverage familiar models and excuses made about the need for speed and the utility of transfer learning. We cannot let volume or nostalgia keep us from taking a sober look at the data and models we currently have, and which ones we actually need to get.
What are the major pain points the biomanufacturing industry is currently facing?
Downstream processing is the work of separating target molecules from the background noise of production. In purely chemical and petrochemical fields, separation processes are well established, extensively characterized, and relatively standardized. This is not the case in industrial biomanufacturing, where upstream flows are arguably more variable and complex than in petrochemicals. Producers on the biomedical side of biomanufacturing who make antibiotics, biologics, and other pharmaceuticals have worked on this problem for a long time. Their products tend to be more expensive and worth specialized handling. The time the field has spent developing the techniques in the urgent pursuit of human health works in their favor for innovation. However, separating fermentation broth from arbitrary commodity molecules is still a major hurdle for a bioindustrial sector already facing so many other simultaneous challenges. Without a robust library of downstream processing methods and a workforce versant in their development and deployment, new industrial products are viewed as significant scaling risks and are funded accordingly.
There is fatigue as well. For the sake of argument, let us peg the onset of the modern era of industrial biomanufacturing to the turn of the latest century. There have been the requisite amount of promises any field must make to build itself into prominence, but there has not been the progress that engenders trust in those or future promises. We have touted synthetic biology as the answer for two and a half decades but our dependence on petroleum for chemicals is as intense as ever. The goodwill we need to shift an entire industry is not a renewable resource. It takes capital, it takes time, and it takes faith that those investments will pay off. But now the chemical companies we need to adopt new solutions have lost some confidence. The policy makers we need to lean into alternative paths and visionary funding are losing trust. If the public from whence government funding ultimately springs descends into skepticism, we may lose our chance to pivot and deliver.
The right investment right now will spell the difference between life and death on this planet for billions of people.
This dangerous dearth of confidence can be addressed by doing something difficult: owning up to it. No one has ever said “oh goody — a chance to do a postmortem!”. But such introspective exercises are critical to making effective changes. A lack of reflection is a tacit vote for the status quo, which is comfortable because we’re rarely punished for a lack of advocacy. We should commission an honest look at the last thirty years — without judgment, without anger, and without the need to reframe disappointing attempts as fractional successes for granting agencies, or position singular successes as broadly representative of progress for egos.
Biomanufacturing is so promising! With proper care and attention it will be incredibly transformative. The right investment right now will spell the difference between life and death on this planet for billions of people. We owe it to ourselves and to science to do it right — which we can only do by acknowledging what we need to change and then truly committing to those changes.
Corn sugar tends to be the most utilized biomass in the bioeconomy. What are the issues the U.S. faces if it continues to rely solely on corn sugar as biomass?
History shows that low-volume, high-margin fine chemicals can be made profitable on corn sugar, but high-volume, low-margin commodity chemicals cannot. Projects that produce fine chemicals and pharmaceuticals see commercial success but suffer from feedstock availability and scaling capacity. Success in high-margin markets encourages people to use the exact same technology to attempt low-margin markets, but then they struggle to reduce costs and improve titers. When a commodity chemical endeavor starts to flag, it can pivot to high-margin markets. This is a pattern we see again and again. As long as corn sugar is the default biomass, it will not change; the United States will not be able to replace petrochemicals with biomanufacturing because the price of corn sugar is too high and cannot be technologically reduced. This pattern is also perpetuated because the yeast we usually ask to do biomanufacturing cannot be made to consume anything but corn sugar. We also struggle to produce arbitrary chemicals in scalable amounts from corn sugar. We are stuck in an unproductive reinforcing spiral.
Even if commodity projects could profit using corn sugar, there is not enough to go around. How much corn sugar would we have to use to replace even a fifth of the volume of petroleum commodity chemicals we currently rely on? How much more land, nitrogen, water, and additional carbon emissions would be needed? Would chemical interests begin to overpower food, medical, and energy interests? What if a pathogen or natural disaster wiped out the corn crop for a year or two? Even if we could succeed at manufacturing commodities with corn sugar alone, locking out alternatives makes the United States supply chain brittle and vulnerable.
MicroByre is working to advance alternatives to substrates currently favored by the bioeconomy.
Continued reliance on corn sugar slows our technological development and stifles innovation. Specialists approaching manufacturing problems in their domain are necessarily forced to adopt the standards of neighboring domains. A chemical engineer is not going to work on separating a biomass into nutrition sources when no microbiologist is offering an organism to adopt it. A molecular biologist is not going to deploy a specialized metabolic pathway dependent on a nutrition source not found in corn sugar. Equipment vendors are not going to design tools at any scale that stray from a market demand overwhelmingly based on the use of corn sugar. Grantors direct funds with the guidance of universities and industry leaders, who are biased towards corn sugar because that’s what they use to generate quick prototypes and spin out new start up companies.
The result of relying on corn sugar is an entrenched field and consequently we might lose our chance to make a difference. Without introducing low-cost, abundant feedstocks like wastes, we run the risk of disqualifying an entire field of innovation.
What does the U.S. need to do in order for other biomass sources to be utilized beyond corn sugar? Are there ideas (or specific programs) that the U.S. government could supercharge?
Federal agencies must stop funding projects that propose to scale familiar yeasts on corn sugars to produce novel industrial chemicals. We must immediately stop funding biomass conversion projects meant to provide refined sugars to such endeavors. And we must stop any notion of dedicating arable land solely to corn sugar solely for the purposes of biomanufacturing new industrial products. The math does not and will not work out. The United States must stop throwing money and support at such things that seem like they ought to succeed any minute now, even though we have been waiting for that success for 50 years without any meaningful changes in the economic analysis or technology available.
Ironically, we need to take a page from the book that cemented petroleum and car supremacy in this country. We need to do the kind of inglorious, overlooked, and subsequently taken for granted survey of the kind that enabled the Eisenhower Interstate System to be built.
We need to characterize all of the non-corn feedstocks and their economic and microbial ecosystems. We need to know how much of each biomass exists, what it is composed of, and who is compiling where. We need to know what organisms rot it and what they produce from it. We need to make all of that data as freely available as possible to lower the barriers of entry for cross-disciplinary teams of researchers and innovators to design and build the logistical, microbiological, chemical, and mechanical infrastructure necessary. We need to prioritize and leverage the complex biomasses that cannot just be ground into yeast food.
We need to get the lay of the land so – to use the roadway analogy – we know where to pour the asphalt. An example of this sort of effort is the Materials Genome Initiative, which is a crosscutting multi-agency initiative for advancing materials and manufacturing technology. (And which has, to my chagrin, stolen the term “genome” for non-biological purposes.) An even more visible example to the public is a resource like the Plant Hardiness Zone Map that provides a basis for agricultural risk assessment to everyone in the country.
The United States needs to lean into an old strength and fund infrastructure that gives all the relevant specialties the ability to collaborate on truly divergent and innovative biomass efforts. The field of industrial biomanufacturing must make a concerted effort to critically examine a history of failed technical investments, shake off the chains of the status quo, and guide us into true innovation. Infrastructure is not the kind of project that yields an immediate return. If venture capital or philanthropy could do it, they would have already. The United States must flex its unique ability to work on a generational investment timeline; to spend money in the very short term on the right things so as to set everyone up for decades of wildly profitable success — and a safer and more livable planet.